INTRODUCTION
Buckwheat, a dicotyledonous angiosperm of Fagopyrum in the Polygonaceae family, is a traditional crop widely planted in the world. The main buckwheat species are common buckwheat (Fagopyrum esculentum Moench) and Tartary buckwheat (Fagopyrum tataricum (L.) Gaertn.) [Kim et al., 2023]. Tartary buckwheat has strong ecological adaptability and can still grow well in harsh environments. It has a balanced amino acid composition, with contents of protein, fat, minerals, vitamins, and trace elements being generally higher than in common buckwheat, wheat, rice, and corn grains [Zhu, 2016]. In addition, it is rich in flavonoids, such as rutin, which are lacking in many cereal crops [Xue et al., 2022]. Tartary buckwheat has been reported to prevent and control diabetes [Wu et al., 2018], lower blood lipids [Sun et al., 2019], lower blood pressure [Hou et al., 2017], as well as elicit anti-cancer [Zhou et al., 2019] and anti-fatigue [Miao et al., 2016] effects. Due to these health-promoting characteristics, Tartary buckwheat is considered a promising raw material for functional food development with both nutritional and bioactive value. At present, it is commonly used in the production of various foods, such as tea, noodles, biscuits, and beverages [Zhu, 2016].
Tartary buckwheat bran (Tbb) is the outermost layer of Tartary buckwheat seeds and is usually a by-product of Tartary buckwheat flour processing. Due to its coarse texture and low digestibility, it is mostly used for feed production, as a fermentation medium or directly discarded, thus not only polluting the environment but also greatly increasing waste resources. However, Tbb is also rich in protein, minerals, and other nutrients and functional compounds [Sinkovič et al., 2022]. Studies have shown that Tbb contains more bioactive compounds than Tartary buckwheat flour [Noda et al., 2023; Xue et al., 2022]. The dietary fiber in Tbb confers important physiological benefits, including the prevention of constipation, reduction of serum cholesterol, and regulation of blood glucose levels [Guo et al., 2012]. As such, Tbb holds considerable potential as a functional food ingredient with broader nutritional and application potential than the flour itself.
Tbb is broadly utilized, particularly in the processing of traditional staple foods like steamed bread, which is a traditional Chinese food with a long history, primarily produced through wheat flour fermentation, dough mixing, and steaming. While steamed bread is known for retaining more nutrients than Western-style baked bread, its widespread consumption has contributed to nutritional imbalances, such as excessive macronutrient intake and insufficient dietary fiber, leading to health issues like coronary heart disease, type 2 diabetes, and obesity [Liu et al., 2011; Rose et al., 2010]. Adding Tbb to wheat flour formulations could help develop bran-enriched steamed bread, offering a nutritionally-enhanced alternative to conventional steamed bread. However, incorporating Tbb into steamed bread presents challenges, including poor taste, altered texture, and darkened color [Ma et al., 2021]. These issues are largely attributed to Tbb’s physicochemical properties, which interfere with the gluten network, slowing fermentation, reducing gas retention, and decreasing loaf volume [Zhao et al., 2025].
To address these challenges, various modification techniques have been explored to improve the sensory and nutritional qualities of Tbb-enriched products. The main methods for modifying bran include fermentation [Coda et al., 2014], superfine grinding [Xiao et al., 2022], extrusion expansion [Li et al., 2023], microwave heating [Jiang et al., 2016], steam explosion [W. Li et al., 2022], ultra-high pressure [Xia & Li, 2018], etc. Despite the widespread application of these techniques to other brans, such as wheat [Guo et al., 2025; Saroj et al., 2025] and barley [Xi et al., 2023; Y. Zhang et al., 2024], research on Tbb modification, especially in the context of steamed bread production, remains limited.
This study aimed to investigate the effects of three modification techniques – fermentation, superfine grinding, and extrusion expansion – on the content of soluble dietary fiber (SDF), total phenolics, and flavonoids in Tbb. The optimal modification method was identified, and fermentation parameters were optimized using single-factor experiments combined with response surface methodology (RSM). Modified Tbb was then incorporated into wheat flour at varying substitution levels (0%, 5%, 10%, 15%, 20%, and 25%, w/w) to produce steamed bread. The resulting products were evaluated in terms of specific volume, textural profile, soluble dietary fiber content, total flavonoid content, and in vitro digestibility. A comprehensive sensory evaluation was also conducted to assess the quality of the bran-enriched steamed bread. This study provides experimental evidence and technological support for the integrated utilization of Tbb and its value-added applications, particularly in improving the quality of traditional steamed bread products.
MATERIALS AND METHODS
Materials
Tartary buckwheat bran powder was from Shanxi Yanmen Qinggao Shiye Co., Ltd. (Shanxi, China), instant active dry yeast was from Angel Yeast Co., Ltd. (Hubei, China), and plant-derived Lactobacillus spp. and Saccharomyces cerevisiae strains were purchased as dried powders from Kunshan Bisour Biotechnology Co., Ltd. (Jiangsu, China) for use in fermentation processes. Standard gallic acid and sodium carbonate were from Sunny Biotech Co., Ltd. (Shanghai, China), Folin-Ciocalteu-phenol reagent and standard rutin were from Hefei Bomei Biotechnology Co., Ltd. (Anhui, China), sodium hydroxide and anhydrous ethanol were from Tianjin Huihang Technology Co., Ltd. (Tianjin, China), sodium nitrite and sodium sulfite were from Tianjin chemical reagent supply and marketing company (Tianjin, China). Potassium sodium tartrate was obtained from Tianjin FengChuan Chemical Reagent Technology Co., Ltd. (Tianjin, China). Aluminum nitrate was obtained from Xilong Scientific Co., Ltd. (Guangdong, China). Golden embryo corn oil was from Shandong Sanxing Corn Industry Technology Co., Ltd. (Shandong, China). Wheat flour was purchased from China Oil and Foodstuffs Corporation (Beijing, China). Saccharifying enzyme (50,000 U/g) and 3,5-dinitrosalicylic acid (DNS) were bought from Shanghai Ruiyong Biotechnology Co., Ltd. (Shanghai, China). Amylase (3,000 U/mL) was from Nanning Pangbo Biological Engineering Co., Ltd. (Guangxi, China). All reagents used were of analytical reagent grade, and the water was ultrapure.
Tartary buckwheat bran modification procedures
Extrusion expansion
The Tbb was processed by using a twin-screw extruder (TSE70, Jinan Sunward Machinery Co., Ltd., Shandong, China). Material moisture content was 18%. The processing parameters were as follows: feeding rate was 15 Hz; screw speed was 200 rpm; and temperature in zone 1, zone 2, and zone 3 was 60°C, 170°C, and 190°C, respectively. After processing, the extrudates were oven-dried at 50°C overnight and then treated in a high-speed multifunctional grinder (Beijing Rayleigh Analytical Instrument Corp., Beijing, China), and sifted through a 100-mesh sieve.
Fermentation
The Tbb powder was mixed with distilled water at a ratio of 1:2 (w/v), after which the mixed culture of S. cerevisiae and Lactobacillus (2:1, w/w) was inoculated into the resulting mixture at an inoculation rate of 3% (w/v). The entire mixture was then incubated at 33°C for 4 h in a constant temperature incubator (GNP-9160, Ningbo Southeast Instrument Co., Ltd., Zhejiang, China). Afterwards, it was dried in a hot air-drying oven (FX101-3, Shanghai Shuli Instrumentation Co., Ltd., Shanghai, China) at 40°C for 48 h, crushed, and sifted through a 100-mesh sieve.
Superfine grinding
The Tbb was put into a superfine grinder (RT-UF26W, Rong Tsong Precision Technology Co., Ltd., Taiwan, China), and the superfine Tbb powder of more than 100 mesh was obtained by using the high-speed airflow generated by the high-speed impact between Tbb and the cutting tool. It was collected according to the centrifugal principle of the cyclone. Processing parameters were as follows: power 1.94 kW, speed 25,000 rpm, and feed size 2 mm.
Determination of contents of functional compounds in Tartary buckwheat bran
Soluble dietary fiber content
The content of soluble dietary fiber (SDF) in Tbb was determined according to the AOAC International method no. 991.43 [AOAC, 2023], and the results were expressed as g of SDF per 100 g of Tbb.
Total phenolic content and total flavonoid content
The extraction of unmodified and modified Tbb was conducted in order to determine the contents of total phenolics and total flavonoids. The powdered samples were treated as follows: 25 mL of ethanol (95%) were added to 1 g of each sample, which was then sonicated at 20°C and 100 W for 60 min, and filtered.
The total phenolic content (TPC) and the total flavonoid content (TFC) were determined by Folin-Ciocalteu colorimetric and aluminum nitrate-sodium nitrite colorimetric methods with some modifications. The TPC of Tbb was determined using the procedure reported by Pirca-Palomino et al. [2024]. To this end, 0.1 mL of the above extract was mixed with 1.0 mL of the Folin–Ciocalteu reagent. After 2 min, 1.5 mL of a sodium carbonate solution was added, the mixture was thoroughly mixed and incubated in the dark for 1 h, and the absorbance was measured at 765 nm using a UV-1800B ultraviolet spectrophotometer (Beijing Rayleigh Analytical Instrument Corp. Beijing, China). Gallic acid was used as the reference substance, the results were expressed as mg gallic acid equivalents (GAE) per g of Tbb. The TFC was determined using the procedure reported by Ge & Wang [2020]. In brief, 1.0 mL of the Tbb extract was mixed with 0.5 mL of a 5% NaNO2 solution and allowed to stand at room temperature for 5 min. Then, 0.5 mL of a 10% Al(NO3)3 solution was added, mixed thoroughly, and the mixture was left to stand for another 5 min. Subsequently, 2 mL of a 4% NaOH solution were added, followed by 60% (v/v) ethanol to bring the final volume to 20 mL. After thorough mixing, the absorbance was measured at 510 nm. The results were expressed as mg rutin equivalents (RE) per g of Tbb.
Determination of processing properties of Tartary buckwheat bran
Determination of water-holding capacity
The water-holding capacity (WHC) was determined based on the procedures described by Sangokunle et al. [2020] and Nguyen et al. [2023], with slight modifications. An accurately weighed modified and unmodified Tbb samples of 1 g (m0) were immersed in 50 mL of distilled water. The mixture was magnetically stirred for 30 min and then allowed to stand at room temperature for additional 30 min. Subsequently, it was centrifuged at 1,800×g for 20 min to remove the supernatant. The remaining residue was collected and weighed as m1. The WHC of Tbb was calculated according to Equation (1):
Determination of oil-holding capacity
The oil-holding capacity (OHC) was evaluated using the procedures described by Sangokunle et al. [2020] and Nguyen et al. [2023], with slight modifications. Both modified and unmodified Tbb samples of 1 g (M0) were immersed in 20 mL of corn oil and magnetically stirred for 30 min. The mixture was then allowed to stand at room temperature for 30 min, followed by centrifugation at 1,800×g for 20 min. After removing the supernatant, any oil adhering to the inner wall of the centrifuge tube was carefully wiped off, and the remaining mass was weighed as M1. The OHC was calculated using Equation (2):
Determination of swelling capacity
The procedures described by Sangokunle et al. [2020] and Nguyen et al. [2023] were used to measure swelling capacity (SC). The samples of modified and unmodified Tbb (m) were immersed in 25-mL graduated cylinder and mixed thoroughly. The initial volume of the sample (V0) was recorded. Subsequently, 10 mL of distilled water were added, and the mixture was shaken well. After standing at room temperature for 24 h, the final volume of the sample (V1) was recorded. The swelling capacity (SC) was calculated using Equation (3):
Optimization of Tartary buckwheat bran fermentation
According to the modification method described above, Tbb was thoroughly mixed with distilled water at a ratio of 1:2 (w/v) to obtain a homogeneous mixture. With the inoculation level of the mixed starter fixed at 3% (w/v), the effects of different ratios of S. cerevisiae to Lactobacillus (1:1, 1:2, 1:3, 2:1, 3:1, w/w), fermentation temperature (23°C, 28°C, 33°C, 37°C, 42°C), and fermentation time (2, 4, 6, 8, 10 h) on the SDF content of Tbb were investigated using a single-factor experimental design in which one variable was changed at a time while the others were kept constant. Specifically, during analyses of the ratio of inoculation cultures, the fermentation temperature and time were maintained at 33°C and 4 h, respectively; during temperature tests, the ratio of inoculation cultures and fermentation time were maintained at 1:1 (w/w) and 4 h, respectively; and during time tests, the ratio of inoculation cultures and fermentation temperature were maintained at 1:1 (w/w) and 33°C, respectively.
According to the design principle of the Box-Behnken central combination experiment, a response surface analysis experiment with three factors and three levels was conducted (Table 1) to optimize the conditions of Tbb fermentation.
Production of Tartary buckwheat bran steamed bread
The mixed flour was prepared by blending fermented Tbb powder and wheat flour. The composite flours were formulated by replacing wheat flour with fermented Tbb at 0, 5, 10, 15, 20, and 25% (w/w, based on total flour). The mixtures were stored in a cool, dry place for later use and the steamed bread was prepared based on the method of S. Zhang et al. [2024] with slight modifications. Specifically, 110 g of the prepared flour was weighed, 1% (w/w) active dry yeast was added, and subsequently 50 g of water were added gradually in multiple portions. All ingredients were mixed in a dough mixer (CE6001C, Guangdong Weishida Electric Technology Co., Ltd., Guangdong, China), first at a low speed for 2 min, then at a medium-high speed for 8 min, to form a smooth dough. The dough was evenly divided into 50-g portions and kneaded into small dough pieces. The dough pieces were placed in a fermentation chamber and proofed for 1 h at 35°C and 60% relative humidity. Subsequently, the samples were steamed for 20 min. After steaming, the heat was turned off, and the samples were allowed to cool naturally at room temperature.
Determination of steamed bread quality
Specific volume
The specific volume measurement was conducted using the rapeseed displacement method outlined by Ouyang et al. [2024]. The cooled steamed bread was placed into a container filled with rapeseed, then the volume of rapeseed displaced was measured with a graduated cylinder. The specific volume was calculated as the ratio of this volume (mL) to the mass of the bread sample (g).
Sensory evaluation
This study was approved by the College of Agronomy and Forestry Science of Hebei North University (Zhangjiakou, China), and informed consent was obtained from all volunteers prior to their participation. Throughout the sensory evaluation, the principles set out in the Declaration of Helsinki were strictly followed. Before the formal assessment, all volunteers attended a brief training session to familiarize themselves with the specific attributes to be evaluated, including appearance, internal structure, color, chewiness, elasticity, odor, and other relevant sensory characteristics. After the steamed bread had been placed at room temperature 25±2°C for 1 h, it was cut into several pieces. Fifty evaluators who met the standards of sensory evaluators both psychologically and physiologically were selected to form a sensory evaluation panel to taste steamed bread. The sensory evaluation included the assessment of several attributes, such as appearance and shape, internal structure, color, chewiness, elasticity, palatability, fragrance, and bitterness. The full score was 100, and the scoring criteria for steamed bread are outlined in Table 2.
Table 2
The scoring standard of steamed bread.
Texture characteristics
The texture characteristics of the steamed bread were studied by texture profile analysis (TPA) with a TA.XT Plus physical property analyzer (Beijing Lotun Science Co., Ltd., Beijing, China). The parameters measured included hardness (N), chewiness (N), springiness (%), cohesiveness, and resilience (%). Springiness was expressed as the percentage recovery of sample height after compression, while resilience was calculated as the percentage of energy recovered during the first compression cycle. After having been left at room temperature for 1 h, the steamed bread was cut into uniform pieces of 15 mm thickness, and the central two pieces were taken for determination. The measurement parameters were set as follows: pretest speed 1.00 mm/s, test speed 5.00 mm/s, post-test speed 5.00 mm/s, compression degree 60.00%, and trigger force 5.0 g.
Determination of contents of functional compounds in steamed bread
The content of SDF in steamed bread was determined according to the AOAC International method no. 991.43 [AOAC, 2023], and the results were expressed as g of SDF per 100 g of dry matter (DM) basis.
Based on the procedures described by Ma et al. [2013], the steamed bread samples were freeze-dried, ground into powder, and passed through a 100-mesh sieve. The powder was extracted with 70% (v/v) ethanol at a solid-to-liquid ratio of 1:20 (w/v) under ultrasonic assistance for 40 min at room temperature. The extract was centrifuged, and the supernatant was collected for TFC determination, which was performed using the above-described method. Rutin was used as the standard, and the results were expressed as mg rutin equivalents per g of bread DM.
Digestibility of steamed bread
To weighed 0.5-g steamed bread crumbs, 10 mL of sodium acetate buffer (0.2 M) were added, and the mixture was placed in a boiling water bath for 30 min. After cooling to room temperature, it was heated in a 37°C water bath (single row of twohole constant temperature water bath pot, Shanghai Shuli Instrumentation Co., Ltd.) for 5 min, and then 1 mL of α-amylase (3,000 U/mL) and 0.1 mL of a saccharifying enzyme (10,000 U/mL) were added. Subsequently, 0.5-mL samples were taken at 0, 20, 60, 90, and 120 min respectively, and 4.5 mL of anhydrous ethanol were added, then the samples were shaken in a constant temperature oscillation incubator several times and centrifuged at 1,800×g for 15 min [Englyst et al., 2018]. The supernatant was collected, and the glucose content in the sample was determined by the DNS method [Miller, 1959]. The digestibility of starch was characterized by the reducing sugar produced in the digestion process.
Statistical analysis
Design-Expert 11 software (Stat-Ease, Inc, Minneapolis, MN, USA) was used for response surface analysis. Microsoft Office Excel 2019 software (Microsoft Corporation, Redmond, WA, USA) was used to calculate means and standard deviations of quality indicators. Data analysis and image processing of the texture analysis were conducted by Texture Exponent 32 software (Beijing Lotun Science Co., Ltd., Beijing, China). Statistical analysis, such as one-way analysis of variance (ANOVA) and Duncan’s multiple comparisons, was performed using SPSS 26.0 software (IBM Corp., Armonk, NY, USA). Differences between means were considered statistically significant at p<0.05. The statistical plots were produced using Origin 2019 software (OriginLab Corporation, Northampton, MA, USA). Each experiment was repeated three times (n=3).
RESULTS AND DISCUSSION
Tartary buckwheat bran modifications
Effect of different modifications on the appearance of Tartary buckwheat bran
Appearance is one of the key indicators directly reflecting product quality. The Tbb appearance before and after modifications is shown in Figure 1. The unmodified Tartary buckwheat bran (Figure 1A) appeared coarse in texture, yellowish in color, and tended to clump into large aggregates. After modification, the extruded bran became dark brown due to exposure to high temperature and pressure, with larger particles and a more coarse, fluffy texture (Figure 1B). In contrast, Tbb modified by fermentation appeared lighter in color and more loosely structured compared to the unmodified version (Figure 1C). The superfine ground Tbb had a brighter and more uniform color, and also finer and more delicate particles (Figure 1D).
Effects of different modifications on the content of functional compounds in Tartary buckwheat bran
The SDF content before and after modifications of Tbb is shown in Figure 2A. The SDF content in all treated Tbb was significantly (p<0.05) higher than in the unmodified one. The SDF content of the unmodified Tartary buckwheat bran was 1.87 g/100 g, and it increased by 106.95%, 59.36%, and 43.85% after extrusion expansion, fermentation, and superfine grinding, respectively. Cao et al. [2021] treated rice bran with a twin-screw extruder, and the results showed that the SDF content of rice bran increased after extrusion, which was consistent with the results of our study. In the extrusion process, the conditions of high temperature, high pressure, and high shear can destroy the dietary fiber molecules and convert insoluble dietary fiber (IDF) into SDF. Zhao et al. [2017] found that the SDF content of wheat bran fermented by yeast and lactic acid bacteria increased. It might be due to the secretion of various enzymes by microorganisms during the growth and reproduction process, which hydrolyzed the protein and starch in the bran. At the same time, some cellulose was hydrolyzed under the action of microorganisms, and IDF was converted into SDF. The reason for the increase in SDF content in the bran after superfine grinding might be that strong friction and impact reduced the particle size of dietary fiber, exposed more functional groups, enhanced the hydrophilicity of dietary fiber, and ultimately increased the content of the soluble components [Zheng et al., 2022].
Figure 2
Soluble dietary fiber content (A), total phenolic content (B), total flavonoid content (C), water-holding capacity (D), oil-holding capacity (E), and swelling capacity (F) of unmodified and modified Tartary buckwheat bran. Different letters above the bars indicate significant differences (p<0.05). GAE, gallic acid equivalent; RE, rutin equivalent.
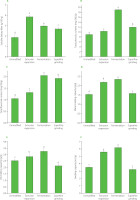
As shown in Figure 2B, the total phenolic content of Tbb increased significantly (p<0.05) after extrusion, fermentation, and superfine grinding treatments. Compared with the unmodified Tbb with a TPC of 8.97 mg GAE/g, the total phenolic content increased by 15.62% after extrusion, by 112.62% after fermentation, and by 32.95% after superfine grinding. The TPC in the fermented Tbb reached 19.07 mg GAE/g.
The total flavonoid content of Tbb is shown in Figure 2C. After extrusion, fermentation, and superfine grinding treatments, it increased significantly (p<0.05), following the trend observed for TPC. Compared with the unmodified Tbb with a TFC of 1.05 mg RE/g, the total flavonoid content increased by 25.26% after extrusion, by 95.25% after fermentation, and by 83.19% after superfine grinding. Fermentation allowed obtaining a Tbb with the total flavonoid content of 2.06 mg RE/g. These results indicate that different modifications effectively promoted the release of soluble dietary fiber and phenolic compounds in Tbb. After extrusion, the TPC of Tbb increased, which was consistent with previous research results; Zhang et al. [2018] found that extrusion increased the free phenolic content of rice bran by 27.1% and decreased the bound phenolics by 27.2%, thereby increasing the total phenolic content of rice bran up to 7.3%. This enhancement may be attributed to involving high temperature and pressure during extrusion, which disrupts the cell wall structure and promotes the release and subsequent extraction of bound phenolic compounds. In the process of fermentation, the TPC and TFC in Tbb increased, this may be attributed to the loosening of the originally dense, cross-linked structure of bran, disruption of cell walls, and release of bound phenolic compounds from the cell wall matrix. This has been demonstrated in studies on wheat bran and Tartary buckwheat bran, where fermentation with lactic acid bacteria alone or in combination with yeasts significantly increased the contents of free phenolic acids and other bioactive components, together with enhanced antioxidant activity [Aung et al., 2022; Tomassi et al., 2025]. In parallel, several reports [Liu et al., 2024; Z.Q. Zhang et al., 2023] have shown that when the raw material was subjected to superfine grinding, the content of extractable phenolics increased markedly. For Tartary buckwheat bran, superfine grinding combined with endogenous enzyme activity substantially modified the flavonoid profile and improved antioxidant and α-glucosidase inhibitory activities [Xiao et al., 2022]. This may be because superfine grinding improves powder uniformity, decreases the average particle size, expands the contact area between phenolics and the extraction solvent, and accelerates their dissolution, thereby increasing the measured phenolic content [Y. Zhang et al., 2023].
Effect of different modifications on the functional properties of Tartary buckwheat bran
As shown in Figure 2D, all used treatments significantly (p<0.05) improved WHC of Tbb. Compared with the unmodified buckwheat bran with a WHC of 1.52 g/g, the corresponding data for the extruded, fermented, and superfine-ground buckwheat bran increased by 42.91%, 52.69%, and 3.67%, respectively. Among them, the fermented bran had the highest WHC of 2.33 g/g.
OHC is an important processing characteristic that can represent the ability of food to absorb and retain fat, improving food flavor and palatability. Compared with the unmodified Tbb, the OHC of Tbb after extrusion and fermentation increased significantly (p<0.05) by 12.40% and 28.19%, respectively (Figure 2E). In contrast, the OHC of Tbb decreased after superfine grinding.
SC of bran affects the texture and processing performance of products. As shown in Figure 2F, compared to the unmodified Tbb, the SC values of Tbb after extrusion and fermentation increased significantly (p<0.05) by 58.28% and 75.24%, respectively. For the superfine-ground Tbb, the SC was 3.38 g/g, being similar (p≥0.05) to that determined for the unmodified Tbb (3.54 g/g).
The results showed that both the WHC, OHC, and SC of Tbb increased after extrusion expansion. This might be due to the relatively increased amount of cellulose and hemicellulose in bran under the action of high temperature, high pressure, and high shear force, with some connection bonds breaking, resulting in more low-molecular-weight compounds and soluble polymers, increasing the specific surface area of the material [Deroover et al., 2020]. The significant improvements in the WHC, OHC, and SC of Tbb after fermentation may be attributed to the degradation of macromolecules, such as IDF, into smaller molecules. Fermentation also increases the specific surface area of SDF, resulting in a loose and porous structure that exposes more polar functional groups [Gu et al., 2020; Y. Li et al., 2022]. These groups can form additional hydrogen bonds or dipole interactions with water, enhancing hydration and water retention. At the same time, more non-polar groups are exposed, which contributes to an increase in OHC. The increase in both polar and non-polar groups facilitates the penetration and tight binding of water and oil into the dietary fiber, thus reducing their loss. After superfine grinding, the WHC increased by 3.67%, whereas the OHC and SC presented an opposite trend compared to WHC, probably because Tbb exposed many hydrophilic groups after superfine grinding, which easily combined with water [Zhao et al., 2009]. At the same time, under the influence of strong mechanical force, macromolecular compounds broke down into smaller particles, and the fiber matrix was destroyed, resulting in a decrease in OHC and SC, which was consistent with the research results reported by Zhu et al. [2010].
In conclusion, extrusion expansion, fermentation, and superfine grinding significantly modified the functional properties of Tbb. Fermentation elicited the most pronounced increase in total phenolic and total flavonoid contents, which are essential for enhancing Tbb’s antioxidant properties – critical attribute for health-focused food development. Furthermore, fermentation offers distinct environmental advantages over extrusion expansion and superfine grinding, which typically demand higher energy inputs and pose greater risk of degradation of heat-sensitive nutrients. Although the SDF content post-fermentation was surpassed by extrusion expansion (Figure 2A), and fermentation yielded superior functional properties of Tbb (Figure 2D–F). Specifically, it achieved the highest WHC, OHC, and SC among the treatments. This combination of enhanced functional properties and superior bioactive compound contents renders fermented Tbb particularly suitable for diverse food applications. Based on these compelling results, fermentation was selected as the preferred modification method for Tbb, and its process parameters were subsequently optimized.
Optimization of Tartary buckwheat bran fermentation process
Single factor experiment
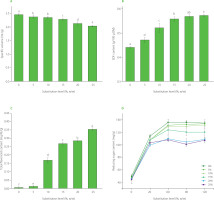
The effect of different ratios of S. cerevisiae to Lactobacillus on SDF content was investigated under the condition of 3% (w/v) addition of inoculation cultures to the mixture of Tbb in water (1:2, w/v) (Figure 3A). In general, Lactobacillus dominate the microbial community during fermentation, creating an acidic environment that severely affects the growth and metabolic activity of S. cerevisiae. On the other hand, an excessive amount of S. cerevisiae can result in over-fermentation, causing the accumulation of metabolic products that inhibit the normal activities of both yeast and lactic acid bacteria. When the ratio of S. cerevisiae to Lactobacillus was 2:1 (w/w), the SDF content of Tbb was the highest, reaching 2.36 g/100 g. Therefore, this ratio of yeast to lactic acid bacteria was used in the next experiment.
Figure 3
Soluble dietary fiber content of Tartary buckwheat bran powders fermented with different ratios of Saccharomyces cerevisiae to Lactobacillus as inoculation cultures (A), at different temperatures (B) and for different times (C). Different letters above the bars indicate significant differences (p<0.05).

The effect of different fermentation temperatures on the SDF content of Tbb is shown in Figure 3B. The SDF content of the bran continuously increased with the increase of fermentation temperature until it peaked at the temperature of 33°C. However, the SDF content decreased significantly after the temperature continued to increase. The results revealed that both high and low temperatures were not conducive to the vital activities of composite microorganisms, affecting the fermentation speed. This might be because when the temperature is too low, the microbial growth is suppressed, making it difficult for yeast and lactic acid bacteria to play a role together. On the contrary, when the temperature is too high, the physiological metabolism of microorganisms is affected. Therefore, the fermentation temperature of 33°C was found optimal.
As shown in Figure 3C, the SDF content presented a trend of first increasing and then decreasing with the prolongation of fermentation time. When the fermentation time was 6 h, the SDF content in the Tbb reached the maximum value of 1.45 g/100 g. This might be because yeast and lactic acid bacteria were still in the growth stage in the initial fermentation phase. As the fermentation time prolonged, the growth of yeast and lactic acid bacteria became more and more stable. When the stable period had been reached, the bacterial yield peaked. As the fermentation time was further prolonged, nutrients were depleted, and SDF was also decomposed and utilized by microorganisms, resulting in a decrease in its content. Therefore, the optimum fermentation time was found at 6 h.
Response surface method optimization experiment
To further optimize the conditions of fermentation, a response surface optimization experiment was designed, as shown in Table 3. Using the SDF content of Tbb as the response, multiple linear regression analysis and quadratic equation fitting were performed. The regression mathematical model for SDF content (Y), fermentation temperature (A), fermentation time (B), and ratio of inoculation cultures (C) was describe by Equation (4):
Table 3
The response surface experiment design scheme and results of optimizing Tartary buckwheat bran fermentation conditions.
The regression model was visualized using the response surface and contour plots (Figure 4). With the increase of fermentation temperature and time, the SDF content showed a trend of first increasing and then decreasing (Figure 4A and B). The slope of the curved surface corresponding to fermentation time was higher than that of the fermentation temperature, indicating that the influence of fermentation time on SDF content was greater than that of fermentation temperature. The contour line was elliptical, indicating a significant interaction between the two factors. Figure 4C and D showed that the SDF content first increased and then decreased with the increase of fermentation temperature and the ratio of inoculation cultures The slope of the curved surface corresponding to the ratio of inoculation cultures was higher than that of the fermentation temperature, indicating that the ratio of inoculation cultures had a stronger effect on the SDF content than fermentation temperature. The contour line was also elliptical, which revealed the significant interaction between the two factors. Figure 4E and 4F showed that with the prolonging fermentation time and the increasing ratio of inoculation cultures the SDF content showed a trend of first increasing and then decreasing. The slope of the curved surface corresponding to the ratio of inoculation cultures was higher than that of fermentation time, indicating that the ratio of inoculation cultures had a stronger effect on the SDF content than the fermentation time. The contour line was also elliptical, and the interaction between these two factors was significant.
Figure 4
Contour (A, C, and E) and response surface (B, D, and F) plots showing the effect of interaction between fermentation temperature and fermentation time (A–B), fermentation temperature and Saccharomyces cerevisiae to Lactobacillus ratio (w/w) (C–D), and fermentation time and Saccharomyces cerevisiae to Lactobacillus ratio (w/w) (E–F) on the soluble dietary fiber content of Tartary buckwheat bran.

According to the model prediction, the optimal conditions for fermentation were: fermentation temperature of 31.93°C, fermentation time of 5.96 h, ratio of yeast to lactic acid bacteria of 53:25 (w/w), and predicted SDF content of 2.97 g/100 g. Based on the operability of the experiment, the final process conditions were set as follows: fermentation temperature of 32°C, fermentation time of 6 h, and the ratio of yeast to lactic acid bacteria of 2:1 (w/w). Three parallel experiments were conducted using these process conditions, and the actual SDF content was determined to be 2.93 g/100 g, which was close to the predicted value, indicating that the process parameters fitted by this model were reliable and had certain practical value.
Effect of different wheat flour substitution levels by fermented Tartary buckwheat bran on steamed bread quality
Sensory evaluation
The sensory scores of steamed breads with different substitution levels of wheat flour by fermented Tbb are shown in Table 4. The sensory score of steamed bread with Tbb increased first and then decreased with an increasing wheat flour substitution level. The highest total score had steamed bread at 10% (w/w) wheat flour substitution by fermented Tbb (84.7). However, when the bran substitution exceeded 10% (w/w), the sensory score decreased significantly (p<0.05), with notable declines in appearance shape, internal structure, color, elasticity, and fragrance. This trend can be attributed to the darkening color caused by flavonoid-rich Tbb. Moreover, Tbb disrupts the gluten network at high substitution levels, reduces dough extensibility, impairs the stability of the gluten network structure, and ultimately results in poor texture [Zhao et al., 2025]. This is similar to the results of the previous study by Zhang et al. [2022], who added different amounts of heat-moisture treated Tbb flour to wheat flour to prepare steamed bread and found that the sensory score first increased and then decreased with the increase of the Tbb flour used.
Table 4
Sensory scores of steamed bread produced with different substitution levels of wheat flour with fermented Tartary buckwheat bran powder.
Texture characteristics
Substituting wheat flour with modified Tbb flour significantly affected the textural parameters of the steamed bread (Table 5). Hardness is an important index to evaluate the quality of steamed bread. As can be seen from Table 5, with increasing wheat flour substitution level, hardness increased steadily from 1,844 N in the control to 3,511 N in the bread with 25% (w/w) Tbb, representing a 90.3% rise (p<0.05). Similarly, stickiness and chewiness increased by 82.0% and 61.6%, respectively, at the highest substitution level. This is primarily due to the high dietary fiber content of Tbb, which competes with gluten proteins for water and interrupts gluten network formation, resulting in a denser and less aerated crumb structure.
Table 5
The texture parameters of steamed bread produced with different substitution levels of wheat flour with fermented Tartary buckwheat bran powder.
Springiness remained relatively stable up to 20% (w/w) Tbb but decreased significantly at 25% (w/w) Tbb (p<0.05). Cohesiveness decreased from 0.90 at 0% to 0.80 at 25% (w/w) Tbb (p<0.05), suggesting a reduction in the bonding strength between gluten proteins. Resilience also decreased significantly (p<0.05), dropping from 0.51 to 0.44 with higher Tbb levels. These reductions indicate that excessive bran incorporation disrupts the continuity of the gluten network, limiting the dough’s ability to recover from deformation and retain gas during proofing [Zhang et al., 2021].
Therefore, when the substitution level was ≤10%, the steamed bread samples exhibited moderate increases in hardness and chewiness while retaining desirable levels of elasticity, cohesiveness, and resilience. Furthermore, they maintained a soft texture, pleasant taste, and appealing appearance. In contrast, higher substitution levels (>15%) led to excessive gluten dilution by bran fibers, resulting in poor gas retention, a denser and drier crumb structure, and significantly reduced sensory acceptance attributable to elevated hardness, loss of elasticity, and a pronounced bitter aftertaste. Thus, 10% wheat flour substitution by fermented Tbb was found optimal for balancing nutritional enhancement with consumer acceptability.
Specific volume
Specific volume is used to reflect the degree of dough volume expansion and retention ability, and is one of the important indicators of steamed bread quality evaluation. As shown in Figure 5A, the specific volume of steamed bread showed an overall decreasing trend with an increasing substitution level. This is consistent with the results of Zhang et al. [2021]. The observed changes might be caused by the fact that the addition of Tbb destroyed the stable gluten network structure in the wheat flour, and the expansion of the steamed bread embryo was blocked, which led to the reduction of steamed bread volume [Li et al., 2023].
Figure 5
The specific volume (A), soluble dietary fiber (SDF) content (B), total flavonoid content (C), and digestive characteristic (D) of steamed bread produced with different substitution levels of wheat flour with fermented Tartary buckwheat bran powder. Different letters above the bars indicate significant differences (p<0.05). RE, rutin equivalent; DM, dry matter.
Internal structure
As shown in Figure 6, the internal pores of the steamed bread without Tbb were uniform but too tight. The pores of the steamed breads with 5% (w/w) and 10% (w/w) substitution levels were even and delicate. When the substitution level exceeded 15% (w/w), with the increase of the Tbb amount, the pores of steamed bread began to vary in size, their number increased, and their internal structure became rougher and rougher.
SDF content
As shown in Figure 5B, compared with the steamed bread without Tbb, the SDF content in the steamed bread increased gradually with an increasing substitution level, and the increase in SDF slowed down when the bran addition amount exceeded 15%.
Total flavonoid content
The wheat flour substitution level with fermented Tbb had significant (p<0.05) effects on the TFC of the steamed bread, which gradually increased with Tbb level increase (Figure 5C). This might be due to the wheat flour substitution with fermented Tbb, which underwent further microbial metabolic activities in the dough, leading to the production of enzymes, such as esterases, glucosidases, and proteases. These enzymes enhanced the breakdown of complex plant compounds, facilitating the release of flavonoids [Huynh et al., 2014]. As the Tbb substitution level increased, the TFC in the steamed bread showed a significant rise. Specifically, at the 5% (w/w) substitution level, the TFC increased 9.38 times compared to the control samples (p<0.05). The increase was particularly pronounced between 5% and 10% (w/w), which represented the most significant interval. Beyond 15% (w/w), the increase in TPC slowed down, indicating a diminishing return with higher substitution levels.
Digestive characteristics
It can be seen from Figure 5D that the reducing sugar content of the steamed bread samples increased first and then stabilized during the whole digestion process. The increase occurred rapidly after the addition of α-amylase within 20 min. Then, the reducing sugars were slowly released and their content slightly increased, reaching a maximum at 60 min. Among all samples, the steamed bread without Tbb exhibited the highest reducing sugar content after digestion, whereas the reducing sugar release decreased progressively with increasing levels of Tbb substitution. This indicates that partial substitution of wheat flour with Tbb significantly reduced starch digestibility. Similar results have been reported in fiber-fortified starch-based foods. Krishnan et al. [2012] demonstrated that the incorporation of wheat bran, oat bran, or rice bran into sweet potato–based pasta significantly slowed starch digestion compared with the control samples. In the present study, the reduced reducing sugar release could be attributed to the higher dietary fiber content in Tbb, which diluted the starch fraction in steamed bread and consequently lowered the amount of digestible substrate available for α-amylase during in vitro digestion [Xue et al., 2022].
CONCLUSIONS
Evaluation of the functional characteristics of Tbb modified by three different techniques showed that extrusion expansion most effectively increased soluble dietary fiber content, while fermentation produced the greatest improvements in total phenolic and total flavonoid contents. Fermentation also significantly improved the WHC, OHC, and SC of Tbb, compared to unmodified Tbb. Collectively, these results indicate that fermentation offers the strongest enhancement of the functional properties of Tbb. These improvements make fermented Tbb a promising ingredient for functional food development. The optimum conditions of fermentation for Tbb were established as: fermentation temperature 32°C, fermentation time 6 h, and the ratio of S. cerevisiae to Lactobacillus as inoculation cultures 2:1 (w/w). The 10% (w/v) substitution of wheat flour by fermented Tbb allowed the production of steam bread with the highest sensory scores. In conclusion, fermentation significantly improved both the functional and processing properties of Tbb, making it a promising ingredient for the development of healthier food products, particularly in the production of traditional steamed bread.



