ABBREVIATIONS
ABTS, 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid); ANOVA, analysis of variance; AOC, antioxidant capacity; C3GE, cyanidin 3-glucoside equivalents; CCRD, central composite rotatable design; CE, catechin equivalents; CV, coefficient of variation; DW, dry weight; FFC, free flavonoid content; FPC, free phenolic content; GAE, gallic acid equivalents; GP, germination percentage; Gt, germination time; [H2O2], hydrogen peroxide concentration; ORAC, oxygen radical absorbance capacity; R2, coefficient of determination; ROS, reactive oxygen species; RSM, response surface methodology; TAC, total anthocyanin content; TE, Trolox equivalents.
INTRODUCTION
Runner bean (Phaseolus coccineus L.), also known as ayocote bean, is indigenous to Mesoamerica and is adaptable to various climatic conditions, particularly in the tropical high-humidity zones of Mexico [Vargas Vázquez et al., 2012]. Although it is a valuable nutritional resource, runner bean remains underutilized as a legume [Alvarado-López et al., 2019]. Traditionally, beans are consumed after cooking [Osuna-Gallardo et al., 2023]. Nevertheless, heightened awareness of health and sustainability issues has spurred interest in alternative methods of consuming legumes and in novel plant-based foods [Aloo et al., 2021].
Sprout production constitutes an effective strategy for diversifying legume consumption [Wojdyło et al., 2020]. Germination can increase the bioactive compound content, such as phenolic acids and flavonoids. These compounds are crucial in treating and preventing chronic degenerative diseases linked to oxidative stress, such as diabetes, hypertension, and cancers [Hernández-Miranda et al., 2025]. Recent research suggests that applying stress during sprout production, particularly during pre-germination stages such as soaking, may enhance the levels of bioactive compounds in germinated legumes [León-López et al., 2020; Yang et al., 2019; Yu et al., 2023]. Nevertheless, the extent of these effects depends on the specific germination conditions [da Silva et al., 2021].
The response surface methodology (RSM) provides a powerful statistical framework for optimizing complex processes such as germination. Within RSM, the central composite rotatable design (CCRD) – which incorporates axial points at lower and upper extremes beyond the factorial range – facilitates accurate response prediction, nonlinear model development, curvature detection, and enhanced model robustness while optimizing multiple parameters with fewer experimental runs and efficient quadratic polynomial fitting [Andres et al., 2020; Mahapatra et al., 2025]. Consequently, this study aimed to evaluate the effects of 24-h soaking with hydrogen peroxide (H2O2) at different concentrations, as well as germination time, on the nutraceutical properties of black runner bean sprouts. The findings provide a basis for developing high-value ingredients for functional foods.
MATERIALS AND METHODS
Plant material
Black runner bean seeds (P. coccineus) used in this study were purchased at the local market in Tecpatán, Chiapas, Mexico, in 2019. The seeds were cleaned and stored in plastic bags at 4°C for subsequent germination assays and chemical determinations.
Runner bean germination process
The germination protocol was adapted from León-López et al. [2020]. Black runner bean seeds were sanitized by immersion in a 0.5% sodium hypochlorite solution (1:5 w/v, ratio) for 10 min. The sanitizing solution was discarded and the seeds were then rinsed three times with purified water. Batches of 30 g of seeds were soaked for 24 h at room temperature with different H2O2 concentrations, as detailed in Table 1. After soaking, the seeds were rinsed with distilled water. The soaked seeds were sown uniformly on filter paper folds placed in plastic containers (20×30 cm). Germination was conducted in an incubator chamber (I-36VL Model, Percival Scientific Inc. Perry, IA, USA) at 25°C and 80–90% of relative humidity until germination time established in the experimental design was reached (Table 1). Germination percentage was calculated at the end of each incubation period. Each assay was performed in quadruplicate. Two replicates were used for physical characterization, while the remaining samples were dried and stored for subsequent chemical analysis.
Table 1
Central composite rotatable design (CCRD) consisting of 13 experiments produced by different combinations of two process variables and five levels: X1 – soaking hydrogen peroxide concentration ([H2O2]) and X2 – germination time (Gt).
Experimental design
The independent variables were: X1 – soaking hydrogen peroxide concentration ([H2O2], 0–35 mM) and X2 – germination time (Gt, 0–96 h). The experimental design employed a CCRD comprising 13 randomized experiments, as detailed in Table 1. These experiments were conducted in random order, and performed in triplicate.
Evaluation of germination percentage
Germination percentage (GP) was evaluated in seeds treated with different H2O2 concentrations and germinated for different times. GP was calculated as the number of successfully germinated seeds, identified by the presence of visible radicles (>1 mm), divided by the total number of seeds sown per treatment, and then multiplied by 100 [León-López et al., 2020].
Estimation of sprout growth
After each germination period, the physical characteristics of the runner bean sprouts were recorded. Radicle length (mm) and radicle diameter (mm) were measured using a digital caliper (CALDI-6MP, Truper. S.A. de C.V., Mexico City, Mexico). The number of seeds that developed secondary roots was recorded to calculate the percentage of secondary root appearance.
Flour obtaining process
The sprouted seeds obtained from each treatment were dried at 55°C for 24 h in a food dehydrator (Hamilton Beach, 32100a, 500 W, Glen Allen, VA, USA). After drying, the sprouts were ground in a coffee grinder (Hamilton Beach, 80350R, Glen Allen, VA, USA). The resulting flours were sifted through an 80-mesh sieve (sieve aperture: 0.180 mm).
Preparation of flour extracts
For optimization purposes, a 0.5 g portion of each flour was extracted by orbital agitation in a horizontal rotary shaker (RKVSD, ART Inc., Laurel, MD, USA) (200 rpm, 25°C) with 4 mL of 80% (v/v) aqueous ethanol during 10 min [Osuna-Gallardo et al., 2023]. The mixture was then centrifuged (Eppendorf 5810R, AG, Hamburg, Germany) at 4,000×g for 10 min, and the supernatant was collected and stored at −20°C for subsequent analysis.
Determination of free phenolic content and free flavonoid content
The free phenolic content (FPC) was determined with the Folin-Ciocalteu colorimetric method of Singleton et al. [1999]. Twenty µL of appropriately diluted extracts obtained as described above were reacted by adding 180 µL of the Folin-Ciocalteu reagent. Following a 20-min incubation, the absorbance of the blue complex was measured at 750 nm using a microplate reader (Synergy HT Multi-Detection BioTek Instruments, Inc., Winooski, VT, USA). Results were calculated using a calibration curve of gallic acid (0–300 mg/L) and presented in mg of gallic acid equivalents (GAE) per 100 g of dry weight (DW) of sprouted seeds (mg GAE/100 g DW).
The free flavonoid content (FFC) was determined using the methodology proposed by Heimler et al. [2005]. In a 96-well plate, 20 μL of the extract were mixed with 100 μL of distilled water and 6 μL of 5% NaNO2, rested for 5 min, then 12 μL of 10% AlCl3 were added. After another 5 min, 40 μL of 1 M NaOH and 22 μL of water were incorporated, followed by a 30-min dark incubation at room temperature. Absorbance was read at 510 nm using a microplate reader (Synergy HT Multi-Detection BioTek Instruments, Inc., Winooski, VT, USA). Results were calculated using a catechin calibration curve (0–300 mg/L) and expressed in mg of catechin equivalents (CE) per 100 g of sprouted seed dry weight (mg CE/100 g DW).
Determination of antioxidant capacity
The antioxidant capacity (AOC) of the sprouted seed flours was evaluated using the 2,2’-azinoazino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) assay [Re et al., 1999]. Each extract (7.5 μL) was added to a 96-well plate and brought to a total volume of 300 μL per well with 292.5 μL of the ABTS•+ solution. The plate was incubated at room temperature for 10 min, followed by absorbance measurement at 735 nm (Synergy HT Multi-Detection BioTek Instruments, Inc., Winooski, VT, USA). Trolox served as the reference standard, with a calibration curve from 0 to 800 µg/mL. Results were expressed as μmol of Trolox equivalents (TE) per 100 g of seed flour on a dry weight basis (μmol TE/100 g DW).
Additionally, the AOC of unprocessed black runner beans and sprouted beans under optimized ([H2O2] of 30 mM, Gt of 92 h) and control ([H2O2] of 0 mM, Gt of 92 h) conditions was determined using the oxygen radical absorbance capacity (ORAC) assay [Ou et al., 2001]. Peroxyl radicals were generated with 2,2’-azobis(2-amidinopropane) dihydrochloride (AAPH), and free radical-induced fluorescence loss was measured in a microplate reader (Synergy HT Multi-Detection, BioTek Instruments, Inc., Winooski, VT, USA) at excitation/emission wavelengths of 485/538 nm. Results were expressed as μmol of Trolox equivalents (TE) per 100 g of seed flour on a dry weight basis (μmol TE/100 g DW).
Regression analysis and optimization
The optimal soaking H2O2 concentration and Gt to maximize GP, FPC, FFC, and AOC in the ABTS assay of sprouted black runner beans were established using the response surface methodology (RSM). The experimental design (CCRD) comprised 13 randomized runs (Table 1), varying [H2O2] (0–35 mM) and Gt (0–96 h). PG, FPC, FFC, and AOC were modelled as quadratic responses according to Equation (1):
where: Y is the predicted response variable (Y1 = PG, Y2 = FPC, Y3 = FFC, Y4 = AOC); βo, βi, βii, and βij are the regression coefficients for the intercept, linear, quadratic, and interaction terms, respectively; Xi and Xj represent the independent variables (X1 = [H2O2], X2 = Gt); and ε denotes the experimental error.
Sprouts characterization
Proximate chemical composition
The moisture (method 925.09), protein (method 920.87), ash (method 923.03), and lipid (method 922.06) contents of black runner beans and sprouted beans under optimized and control conditions were determined according to the official AOAC International methods [AOAC, 2005]. The carbohydrate content was calculated by subtracting the sum of moisture, protein, fat, and ash from 100 g of sample. All results were expressed in g/100 g DW.
Total anthocyanin content
The total anthocyanin content (TAC) was determined after extraction of a weighed 0.1 g portion of flours of unsprouted, control sprouted, and optimal sprouted beans into a 2 mL vial with 1 mL of acidified methanol (95% methanol and 1 M HCl, 85:15, v/v). The samples were vortexed for 10 min (Genie 2 mixer, model G560, Scientific Industries, Inc., Bohemia, NY, USA) and then centrifuged (Eppendorf 5810R, AG, Hamburg, Germany) at 3,000×g for 10 min, and the supernatants were collected. Sample absorbance was immediately recorded at 535 nm (A535) and 700 nm (A700) using a microplate reader (Synergy HT Multi-Detection BioTek Instruments, Inc., Winooski, VT, USA). TAC was quantified using the following Equation (2) [Abdel-Aal & Huel, 1999]:
where: ε is cyanidin 3-glucoside molar absorption coefficient (23,900 L/(cm×mol), Vextract is total extract volume (L), MW is cyanidin 3-glucoside molecular weight (449.2 g/mol), and wsample is weight of the sample (g).
Results were expressed as mg of cyanidin 3-glucoside equivalents (C3GE) per 100 g of sprouted bean dry weight (C3GE/100 g DW).
Composition of free and bound phenolic fractions
The free phenolic fraction was obtained as described in “Preparation of flour extracts” section. The bound phenolics were extracted from the precipitate remaining after free phenolic extraction [Mora-Rochín et al., 2010]. The precipitate was treated with 10 mL of 2 M NaOH, heated at 95°C for 30 min, and stirred for 1 h at room temperature. The mixture was neutralized with 2 mL concentrated HCl, vortexed for 2 min, and defatted by adding 5 mL of hexane. The defatted fraction was then extracted four times with 5 mL of ethyl acetate each time (vortexed for 10 min and centrifuged at 3,000×g for 10 min per extraction). The combined ethyl acetate fractions were evaporated to dryness under reduced pressure in a Speed Vac Concentrator SC 250 EXP (Thermo Scientific Inc., Sunnyvale, CA, USA), and reconstituted in 1 mL of 50% methanol.
The phenolic compound profile of the free and bound phenolic fractions was determined using a Dionex UltiMate 3000 high-performance liquid chromatography (HPLC) system with a photodiode array detector (DAD3000) (Thermo Fisher Scientific, New York, NY, USA) according to the procedure previously used by Valdez-Morales et al. [2014]. The injection volume was 10 µL. Separation was performed on a C18 Acclaim 120 Å analytical column (C18, 5 μm, 120 Å, 4.6×250 mm) from Dionex (Thermo Fisher Scientific, New York, NY, USA), at room temperature using a gradient elution of acetic acid–acidified water (pH 2.8) (A) and acetonitrile (B). The gradient program varied the proportion of solvents over 45 min as follows: 95% A (0–8 min); 6–12% B (8–14 min); 12–20% B (14–18 min); 20–35% B (18–24 min); 35–95% B (24–27 min); 95–60% B (27–31 min); 60–40% B (31–34 min); 40–20% B (34–38 min); and 20–5% B (38–45 min). The flow rate was 0.5 mL/min. Detection was set at wavelengths of 280, 320, and 360 nm. Chromatographic peaks were identified by comparing their retention times and UV-Vis spectra with those of authentic standards. Samples were injected in triplicate, and data were analyzed using Chromeleon 7.0.200 software (Thermo Fisher Scientific, Sunnyvale, CA, USA). Results were expressed as µg/g seed flour DW.
Statistical analysis
The effect of H2O2 treatment on seedling growth, chemical composition, AOC, free phenolic content, free flavonoid content, and phenolic profile was evaluated using a one-way analysis of variance (ANOVA). Mean comparisons were performed with Tukey’s test at the 95% confidence level (p<0.05).
RESULTS AND DISCUSSION
Effect of the soaking with H2O2 and germination time on the growth performance of runner bean sprouts
Radicle length, diameter, and the number of secondary roots were measured in sprouts as outlined in the experimental design (Table 1). Elevated H2O2 concentrations consistently promoted radicle elongation at both 48 h and 82 h of germination (Table 2, Figure 1). At 48 h, radicle length increased from 17.3 mm (0 mM H2O2) to 23.9 mm (35 mM H2O2). At 82 h, values ranged from 41.0 mm (5 mM H2O2) to 46.3 mm (30 mM H2O2). The effect was more pronounced at 48 h, highlighting that H2O2 substantially enhances early root development in runner bean sprouts. No root growth was detected with germination times shorter than 14 h, regardless of H2O2 treatment. Secondary roots were absent at short Gt. At 48 h, secondary root formation was not induced irrespective of the H2O2 concentration applied, and at 82 h, 30 mM H2O2 increased secondary roots by 36 percentage points compared to 5 mM. The presence of secondary roots was primarily determined by Gt and further influenced by H2O2 treatment at high concentrations. These results emphasize the importance of optimizing both Gt and H2O2 concentration to maximize root development.
Table 2
Length and diameter of radicles, and percentage of seeds that developed secondary roots after soaking runner bean in an H2O2 solution in different concentrations ([H2O2]) and germinating for different times (Gt).
| No.1 | [H2O2] (mM) | Gt (h) | Length (mm) | Diameter (mm) | Secondary roots (%) |
|---|---|---|---|---|---|
| 1 | 17.5 | 0 | 0.0 | 0.0 | NP |
| 2 | 5 | 14 | 0.0 | 0.0 | NP |
| 3 | 30 | 14 | 0.0 | 0.0 | NP |
| 4 | 0 | 48 | 17.3 | 2.1 | NP |
| 5* | 17.5 | 48 | 18.9 | 2.3 | NP |
| 6 | 35 | 48 | 23.9 | 2.5 | NP |
| 7 | 5 | 82 | 41.0 | 3.0 | 14.5 |
| 8 | 30 | 82 | 46.3 | 3.4 | 50.5 |
| 9 | 17.5 | 96 | 45.8 | 3.1 | 56.9 |
Figure 1
Appearance of runner bean sprouts at different germination times (48–82 h) after soaking in H2O2 solutions of varying concentrations (0–35 mM).

Previous studies have shown that exogenous H2O2, applied at optimal concentrations, promotes adventitious root formation and elongation across various plant species [Li & Jia, 2013; Roussos, 2023]. In accordance with these findings, Barba-Espin et al. [2010] reported that pea seeds imbibed with H2O2 at concentrations of 0, 5, 10, and 20 mM exhibited a clear stimulation of seedling length, with the highest concentration showing the most pronounced effect. Exogenous H2O2 at suitable doses acts as a priming agent, boosting seedling vigor while limiting oxidative damage [Wojtyla et al., 2016]. Reactive oxygen species (ROS), particularly H2O2, are key signaling molecules in seed physiology, shifting the view from harmful by-products of aerobic respiration to regulators of developmental transitions. During imbibition, H2O2 accumulates through mitochondrial, peroxisomal, and nicotinamide adenine dinucleotide phosphate oxidase activity, facilitating endosperm weakening and hormonal modulation by enhancing gibberellin biosynthesis while reducing abscisic acid and ethylene levels [Černý et al., 2018]. In cereals, H2O2 also promotes α-amylase activation and programmed cell death in the aleurone layer via DELLA protein interactions, processes essential for nutrient mobilization [Nazir et al., 2020]. In turn, antioxidant systems, both enzymatic and non-enzymatic, balance oxidative stress while permitting H2O2 to act as a signaling molecule [Chu et al., 2022]. Moreover, nuclear accumulation of H2O2 at radicle protrusion may regulate redox-sensitive transcription factors, leading to gene expression changes linked to phytohormone signaling [Wang et al., 2025]. Collectively, these findings highlight the multifaceted role of H2O2 in coordinating germination success and phytochemical outcomes.
Mathematical models of response variables
As shown in Table 3, the process variables, i.e., [H2O2] and Gt, significantly influenced the response variables, including GP, FPC, FFC, and AOC. Using multiple regression analysis, quadratic polynomial equations for each response variable were accurately fitted to the experimental data of germination conditions. The response surfaces and the obtained contour plots were then analyzed to visualize these effects (Figure 2). The validity and adequacy of the predictive models were confirmed by considering the statistical parameters, including a high coefficient of determination (R2) and adjusted R2 (>0.80 for both), a very low p-value (<0.05), a coefficient of variation (CV) below 10%, and a non-significant lack-of-fit test (p>0.05) (Table 4).
Table 3
Experimental rotatable central composite design used to obtain treatment combinations of different concentrations of H2O2 ([H2O2]) and germination time (Gt) for producing runner bean sprouts, along with the experimental values obtained for the selected response variables.
[i] 1Does not correspond to order of the experiments. GP, germination percentage; FPC, free phenolic content; FFC, free flavonoid content; AOC, antioxidant capacity in ABTS assay; GAE, gallic acid equivalent; CE, catechin equivalent; TE, Trolox equivalent, DW, dry weight. Values in bold mean maximum values.
Table 4
Parameters of predicted quadratic polynomial model of regression analysis for the response variables including germination percentage (GP), free phenolic content (FPC), free flavonoid content (FFP), and antioxidant capacity (AOC) after adjusting the experimental data.
Figure 2
Response surface and contour plots for the effect of the soaking hydrogen peroxide concentration (H2O2), and germination time (Gt) on the response variables: (A) germination percentage (GP), (B) free phenolic content (FPC), (C) free flavonoid content (FFC), and (D) antioxidant capacity (AOC) of black runner bean sprouts.

Germination percentage
The process variables, [H2O2] and Gt, affected the germination performance in black runner bean sprouts, with GP ranging from 14.0 to 98.0%, depending on the experimental conditions (Table 3). The analysis of variance yielded a significant quadratic model for GP (p<0.0001), which includes the linear terms of [H2O2] and Gt, the quadratic term Gt (Gt2), and the interaction between both variables ([H2O2]×Gt) (Table 4).
The contour plot (Figure 2A) shows a clear GP increase with longer Gt. The treatment with 35 mM H2O2 for 48 h resulted in GP values similar to those achieved after more than 82 h at lower H2O2 concentrations, suggesting that oxidative elicitation expedited germination. The highest GP was observed at H2O2 concentrations above 30 mM combined with Gt exceeding 48 h. Comparable responses have been documented in other legumes treated with H2O2 during imbibition [Barba-Espín et al., 2012; León-López et al., 2020; Santhy et al., 2014]. This behavior is explained by the inductive effect of H2O2 in breaking seed dormancy and increasing germination. The oxidative stress caused by the natural accumulation of H2O2 enhances the production of ROS that diffuse from the seed surface to its interior and, by interacting with other molecules, inhibit the action of abscisic acid cytokinins, and indole-3-acetic acid, while simultaneously increasing the biosynthesis and inhibiting the catabolism of gibberellic acid, thereby promoting the germination process [Delis-Hechavarría et al., 2021; Wojtyla et al., 2016].
Free phenolic content
The FPC in black runner bean sprouts was affected by the process variables, ([H2O2] and Gt), showing values ranging from 46.9 to 71.8 mg GAE/100 g DW across the 13 treatments (Table 3). The analysis of variance yielded a significant quadratic model for FPC (p<0.0001), which includes the linear and quadratic terms of [H2O2] and Gt, and the interaction between both variables ([H2O2]×Gt) (Table 4).
The contour plot (Figure 2B) shows the interaction of the process variables [H2O2] and Gt with FPC in runner bean sprouts, where increasing [H2O2] resulted in the highest FPC. A similar trend was observed with increasing Gt; however, the stimuli caused by the chemical stress induced by different H2O2 concentrations (0, 17.5, and 35 mM) at the same Gt (48 h) showed significant increases in FPC. The highest FPC (71.8 mg GAE/100 g DW) was obtained at Gt of 96 h and [H2O2] of 17.5 mM (Table 3), demonstrating that the synergy between high peroxide concentrations and prolonged germination times maximized FPC in black runner bean sprouts. Previous studies have reported similar positive effects of germination time and H2O2 stress on total phenolic content in wheat sprouts, Ficus deltoidea Jack plant, and chia sprouts, supporting the present findings [Dziki et al. 2015; Nurnaeimah et al., 2020; Gómez-Velázquez et al., 2021]. Dziki et al. [2015] evaluated the influence of germination time (2, 4, 6, and 8 days) on the total phenolic compound content in wheat sprouts, finding that the highest values were obtained at 8 days of germination. On the other hand, several authors observed the same positive effect when stressing seeds or plants with H2O2, finding that concentrations of 16 and 30 mM improved the total phenolic content in ethanolic extracts of F. deltoidea [Nurnaeimah et al., 2020], and concentrations of 10 and 20 mM significantly increased phenolic content in chia sprouts (Salvia hispanica L.) [Gómez-Velázquez et al., 2021].
The observed increase in FPC in H2O2-treated sprouts may result from de novo synthesis and metabolic transformation. Enzymes such as l-tyrosine ammonialyase and l-phenylalanine ammonialyase are highly responsive to both germination duration and H2O2 elicitation [Świeca, 2016]. Additionally, seeds can upregulate defense enzymes like peroxidase and polyphenol oxidase to manage the rapid increase in ROS under stress conditions [Nurnaeimah et al., 2020].
Free flavonoid content
The free flavonoid content in black runner bean sprouts was affected by the H2O2 soaking concentration and Gt showing values ranging from 15.7 to 28.3 mg CE/100 g DW (Table 3). The analysis of variance yielded a significant quadratic model for FFC, which includes the linear terms of [H2O2] and Gt, the quadratic terms of [H2O2], and the interaction term of both variables ([H2O2]×Gt) (Table 4).
The response surface (Figure 2C) shows a progressive increase in FFC with increasing Gt, reaching a maximum at the longest durations tested. H2O2 concentration exerted a secondary but meaningful effect, enhancing flavonoid accumulation at intermediate doses, particularly in combination with extended Gt. These findings indicate that flavonoid biosynthesis is primarily regulated by developmental processes associated with germination and is further amplified by oxidative signals. H2O2 functions as a signaling molecule that modulates the expression of genes involved in flavonoid biosynthetic pathways, while prolonged germination supports sustained metabolic activity and substrate availability. Comparable responses have been reported in Dalia bean [Mendoza-Sánchez et al., 2016], quinoa [Świeca, 2016], F. deltoidea [Nurnaeimah et al., 2020], and chia seeds [Gómez-Velázquez et al., 2021], demonstrating the robustness of H2O2 elicitation across various plants.
Antioxidant capacity
The AOC in black runner bean sprouts ranged from 1,980 to 3,526 μmol TE/100 g DW and was affected by both process variables (Table 3). The analysis of variance yielded a significant two-factor interaction model for AOC, which includes the linear terms of [H2O2] and Gt, as well as the interaction of both variables ([H2O2]×Gt) (Table 4).
The contour plot (Figure 2D) shows that AOC increased as both H2O2 concentration and Gt rose, mirroring the trends observed for FPC and FFC. Notably, the highest AOC value (3,526 μmol TE/100 g DW) was achieved by soaking seeds in 30 mM H2O2 and germinating for 82 h, indicating that high peroxide concentrations can reduce the time required to reach near-maximum AOC, whereas at shorter germination times (e.g., 14 h) peroxide concentration had little effect on AOC. The observed increase in AOC during germination is primarily attributed to elevated levels of phenolic compounds, as the FPC and FFC significantly correlated with AOC.
In line with previous studies on amaranth, lentil, Dalia bean, and chia sprouts [Gómez-Velázquez et al., 2021; Mendoza-Sánchez et al., 2016; Perales-Sánchez et al., 2014; Świeca, 2015], these findings suggest that flavonoids and other phenolics enhanced by H2O2 treatment are key contributors to the improved antioxidant potential of black runner bean sprouts.
Optimization of hydrogen peroxide soaking concentration and germination time
For optimizing process conditions, the RSM was used through the graphical method to determine the optimal combination of the [H2O2] and Gt that maximize the values of the GP, FPC, FFC, and AOC in the sprouts. The overlaid contour plot (Figure 3) was used to determine the best combinations of the process variables. The dark green area shows the region with the best combination of process variables to achieve the highest levels of the four response variables, from which the optimal conditions of [H2O2] (30 mM) and Gt (92 h) were selected.
Figure 3
Optimization graph for the response variables: germination percentage (GP), free phenolic content (FPC), free flavonoid content (FFC), and antioxidant capacity (AOC) of black runner bean sprouts. X1, soaking hydrogen peroxide concentration (H2O2); X2, Germination time (Gt).
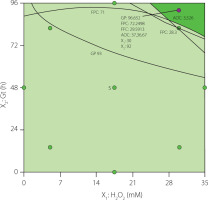
The predicted values at the optimal point under the aforementioned conditions were: GP=96.7%, FPC=72.3 mg GAE/100 g DW, FFC=29.6 mg CE/100 g DW, and AOC=3,737 μmol TE/100 g DW. To validate the accuracy of the prediction model, runner bean seeds were germinated under the optimal conditions, and the response variables GP, FPC, FFC, and AOC were determined. Results are shown in Table 5. Growth performance and proximate chemical composition of sprouts obtained under optimal conditions were also analyzed.
Table 5
Proximate composition, phenolic content, and antioxidant capacity of unsprouted runner bean and sprouted under control and optimal conditions, as well as growth performance of sprouts.
[i] Different lowercase letter superscripts in the same row show significant difference (p<0.05). ABTS assay, assay with 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid); ORAC, oxygen radical absorbance capacity; GAE, gallic acid equivalent; CE, catechin equivalent; TE, C3GE, cyanidin 3-glucoside equivalent; Trolox equivalent, DW, dry weight.
Effect of the optimal treatment on growth performance and proximate chemical composition of runner bean sprouts
To evaluate the effect of soaking treatment with H2O2 on the growth of runner bean sprouts, radicle length and diameter, as well as the percentage of secondary roots, were analyzed. Sprouts obtained under optimal conditions were compared with those obtained by soaking without H2O2 for 92 h (control sprouts). The radicle length in the optimal and control treatments differed significantly (p<0.05), with the former having a 32.0% higher value (Table 5). This indicates a positive trend in the development of structures in runner bean sprouts due to the inductive effect of the H2O2 treatment. However, the radicle’s diameter did not show significant differences (p≥0.05) between the two treatments. Regarding the percentage of seeds with secondary roots, the optimal treatment registered a higher percentage compared to the treatment without the stressor, with 76.7% and 43.0%, respectively, showing a difference, which suggests that H2O2 accelerates the emergence of lateral structures in the seed’s root system. The root is a fundamental organ whose main function is to absorb water and minerals; therefore, a greater length implies a higher probability of success for the establishment, development, and survival of the seedling. Interestingly, Saleh et al. [2019] found a correlation between the increase in their length and the rise in antioxidant capacity as well as the content of phenolic compounds and flavonoids in different legumes. Other authors have observed the same inductive trend of H2O2 on root growth in various seeds [Barba-Espín et al., 2012; Chaplygina et al., 2020]. These results could be explained by the interaction between the redox state and plant hormones orchestrated by H2O2 in the induction of proteins related to signaling and plant development during the early growth of seedlings [Barba-Espin et al., 2010]. In addition, the natural accumulation of H2O2 promotes the energy metabolism required for the growth of the radicle and plumule rather than promoting water absorption in the early stage of germination through an increase in osmotic regulators; it mobilizes sugar reserves derived from stored starch, inhibits catabolism, and promotes the biosynthesis of gibberellic acid, a growth and development regulatory substance in seedlings [Delis-Hechavarría et al., 2021; Song et al., 2023].
The unsprouted runner black beans contained 18.96 g of protein, 2.25 g of lipids, 5.50 g of ash, and 73.29 g of total carbohydrates in 100 g DW (Table 5). This proximate composition largely coincides with those reported by various authors for different bean varieties [Alvarado-López et al., 2019; Corzo-Ríos et al., 2020; Osuna-Gallardo et al., 2023], with minimal variations that could be explained by environmental conditions during cultivation and harvest, the grain variety, and the methodology used. The protein and lipid content in the sprouts obtained under optimal conditions and control sprouts showed significant increases (p<0.05) compared to the unsprouted seeds (Table 5). This increase can be attributed to the loss of dry weight due to the oxidation of carbohydrates during seed respiration and the activation of certain enzymes during the germination process [Nazih et al., 2025]. No significant difference (p≥0.05) was found between the proximate chemical composition of the control sprouted seeds and the seeds sprouted under optimized conditions, contrary to what was reported by León-López et al. [2020], who reported a significant increase in protein content in germinated chickpea grains under chemical stress with H2O2.
Effect of the optimal treatment on phytochemicals and antioxidant properties of runner bean sprouts
The free phenolic content measured in unsprouted runner bean (58.8 mg GAE/100 g DW) was consistent with values reported by Osuna-Gallardo et al. [2023], but lower than those observed in other P. coccineus varieties [Alvarado-López et al., 2019]. Germination resulted in significant (p<0.05) increases in FPC for both the optimal (to 123.7%) and control (to 115.0%) treatments compared to the unsprouted seeds (Table 5). Additionally, the optimal H2O2 treatment yielded significantly (p<0.05) higher FPC than the control, supporting findings in other seeds or grains, including chickpea, chia, and barley, where H2O2 elicitation promoted phenolic accumulation [Delis-Hechavarría et al., 2021; Gómez-Velázquez et al., 2021; León-López et al., 2020]. A higher phenolic content is noteworthy, as these compounds contribute substantially to the antioxidant activity of sprouts.
The free flavonoid content also increased significantly (p<0.05) in both optimal (to 128.1%) and control (to 118.1%) treatments relative to the unsprouted seeds (Table 5). The increase in FFC under the optimal H2O2 treatment is consistent with previous studies in other species exposed to hydroxyl peroxide elicitation [Gómez-Velázquez et al., 2021; Uchegbu & Amulu, 2015], highlighting the role of H2O2 as an effective stressor to enhance flavonoid biosynthesis.
For total anthocyanin content, the value obtained in this study (9.5 mg C3GE/100 g DW) was lower than those previously reported for bean of some P. coccineus varieties [Alvarado-López et al., 2019], but higher than those observed by Osuna-Gallardo et al. [2023], who reported values close to 4 mg C3GE/100 g for unprocessed runner bean. In our study, germination led to a significant (p<0.05) reduction in TAC in both optimal and control treatments compared to the unsprouted seeds (Table 5). James et al. [2020] reported a decrease in anthocyanin content as germination time increased in different legumes, probably because various enzyme systems are mobilized and activated during germination, leading to anthocyanin loss through oxidation and leaching. Meanwhile, Dueñas et al. [2006] evaluated the effect of germination and elicitation on the phenolic profile of bean seeds (Phaseolus vulgaris L.) germinated for 8 days and observed a reduction in total and some specific anthocyanins, reporting that germination in the presence of inducers or stressors caused a more extensive decrease in anthocyanin content even below the quantification limit. The behavior recorded in the present study for the anthocyanin content in black runner bean soaking under the induction of chemical stress with H2O2 is in agreement with these findings, since a greater reduction was observed in the optimal germination treatment than in the control one (Table 5).
The antioxidant capacity (AOC) of unsprouted seeds and sprouts germinated under both optimal and control conditions was evaluated as ABTS•+ scavenging activity and ORAC. According to Munteanu & Apetrei [2021], the ABTS assay operates via a mixed mechanism (involving both electron and hydrogen atom transfer), whereas the ORAC assay relies primarily on hydrogen-atom-transfer mechanisms. The use of both methods provided a more comprehensive assessment of antioxidant properties. The value obtained by the ABTS assay for the unsprouted seed (2,472 μmol TE/100 g DW) was consistent with that reported by Osuna-Gallardo et al. [2023], who reported 2,657.94 μmol TE/100 g of unprocessed runner bean. However, it was higher than the values reported by Orak et al. [2016] for ten white bean (P. vulgaris) varieties (350–517 μmol TE/100 g), and also higher than those reported by Weidner et al. [2018] for four P. vulgaris varieties (421–640 μmol TE/100 g DW). Optimal germination treatment and the control germination treatment showed significant increases (p<0.05) of 53.0% and 22.5%, respectively, compared with the unsprouted bean (Table 5). Likewise, a significant increase (p<0.05) of 24.9% was observed as a result of the soaking treatment with H2O2 when comparing the optimal sprouts to the controls. This increase in the AOC in the sample germinated under optimal conditions could be associated with the stimulation of the synthesis of compounds with high antioxidant activity, such as phenolic compounds, due to the chemical stress produced by the application of H2O2 during germination [León-López et al., 2020].
Several authors have emphasized the increase in AOC during the germination process using H2O2 as an inducing stress agent. For instance, Gómez-Velázquez et al. [2021] recorded an increase in the AOC of chia seeds germinated with 10, 20, and 30 mM H2O2, obtaining increases in the range of 29–37% compared to seeds germinated without the stressor. Similarly, León-López et al. [2020] reported that in white chickpea germination, chemical H2O2 elicitation produced an increase of 14.8% when comparing their optimal treatment ([H2O2] of 30 mM, Gt of 72 h) with control seeds without elicitor ([H2O2] of 0 mM, Gt of 72 h). Conclusions from these studies agree with the present findings regarding the increase in AOC measured by the ABTS assay in runner bean sprouts treated with H2O2.
The ORAC showed the same trend as that determined by ABTS assay (Table 5). Optimal and control germination treatments showed significant increases (p< 0.05) of 31.9% and 16.3%, respectively, compared with ungerminated runner beans, moreover, a significant increase (p<0.05) of 13.5% was observed between runner beans germinated under optimal conditions and those germinated under control conditions. The ORAC of the ungerminated seed (4,982 μmol TE/100 g DW) was close to that reported by Alvarado-López et al. [2019], who reported values of 5,162; 3,694; 2,557; and 2,031 μmol TE/100 g, for four varieties of P. coccineus bean: purple, black, brown, and white, respectively. However, Osuna-Gallardo et al. [2023] recorded an ORAC value of 3,866 μmol TE/100 g of runner bean flour, which was lower than those reported in this study. These variations may be associated with differences in the phenolic profiles among bean varieties, as well as with the extraction and quantification techniques used.
In summary, these results demonstrate that H2O2 treatment during soaking significantly enhanced the antioxidant capacity of black runner bean sprouts, supporting their potential use as nutraceutical ingredients with improved health-promoting properties.
A variety of phenolic acids, flavonoids, and anthocyanins have been reported in runner bean varieties [Baeza-Jiménez & López-Martínez, 2024; López-Martínez, 2020]. Particularly, gallic acid, sinapic acid, ferulic acid, chlorogenic acid, p-coumaric acid, protocatechuic acid, and 4-hydroxybenzoic acid have been identified in unprocessed black runner beans [Baeza-Jiménez & López-Martínez, 2024]. Flavonoids, such as kaempferol 3-glucoside, catechin, and epicatechin, were also reported in unprocessed black runner beans. In this study, gallic, syringic, ferulic, chlorogenic, and p-coumaric acids were identified in unsprouted runner beans (Table 6). Regarding flavonoids, consistent with Baeza-Jiménez & López-Martínez [2024], catechin and quercetin were identified; in addition, rutin was detected in the free phenolic fraction of the unsprouted seeds. Germination processes can modify the phenolic profile [Domínguez-Arispuro et al., 2018; Yu et al., 2023], and even soaking stress can elicit an increase in some phenolic contents [León-López et al., 2020].
Table 6
Phenolic compound profile of unsprouted runner bean and sprouted under control and optimal conditions (µg/g dry weight).
Among the main findings, the significant (p<0.05) increase in the content of gallic acid and syringic acid, and the presence of caffeic acid, stand out as a result of germination in the free phenolic fraction (Table 6). The content of catechin and quercetin also significantly increased (p<0.05) in the free phenolic fraction as a result of the control germination treatment. The stress treatment with H2O2 during soaking induced specific changes in the free phenolic fraction compared to the unprocessed seed and the control sprouts; notably, a significant increase in ferulic acid and p-coumaric acid was observed. In contrast, the content of catechin and quercetin decreased significantly (p<0.05) compared to the control sprouts.
Although the content of bound phenolic compounds was not used as a response variable for the RSM optimization, nor were their antioxidant properties individually measured, it was considered important to analyze the phenolic profile of this fraction to fully discuss the metabolic changes that occurred. Ferulic and p-coumaric acids, which increased in both free and bound phenolic fractions in runner bean sprouts obtained under optimal conditions, serve as potent antioxidants with applications across food, nutraceuticals, and pharmaceuticals. Ferulic acid supports metabolic health by enhancing glucose and lipid metabolism and mitigating oxidative stress, while also exerting anti-inflammatory, antimicrobial, neuroprotective, and cardiovascular effects [Jacobo-Velázquez, 2025; Kumar et al., 2025]. Meanwhile, p-coumaric acid provides anti-inflammatory, antidiabetic, anticancer, cardioprotective, and hepatoprotective benefits, positioning both phenolic acids as promising candidates for drug formulations and chronic disease prevention [Kaur & Kaur, 2022; Kumar et al., 2025].
CONCLUSIONS
The present study demonstrates that H2O2 treatment before black runner bean seed germination is an effective strategy to enhance sprout quality. H2O2 elicitation improved germination performance, stimulated radicle growth, increased the accumulation of total phenolic compounds, and enhanced the antioxidant capacity of sprouts. Optimal conditions, 30 mM H2O2 combined with prolonged germination (around 92 h), resulted in higher levels of certain free and bound phenolic acids and flavonoids, as well as improved ABTS•+ scavenging activity and ORAC of the sprouts. The use of response surface methodology (RSM) was critical for identifying these optimal elicitation conditions, offering a robust statistical framework that facilitates reproducibility and potential scale-up. These findings highlight the promise of H2O2-assisted sprouting for the development of functional flours with enhanced nutritional and potential health benefits.

