INTRODUCTION
Beta vulgaris L. is a plant of considerable economic and nutritional importance, comprising four main cultivated forms: red beet, Swiss chard, fodder beet, and sugar beet [Mroczek, 2015]. It is a rich source of bioactive compounds, including polyphenols, betalains, and triterpene saponins, which are increasingly recognized for their health-promoting and disease-preventive properties [Biswas & Dwivedi, 2019]. Among these compounds, triterpene saponins have attracted considerable attention owing to the broad range of their biological activities, including antiviral, anticancer, antimicrobial, anti-inflammatory, antihypertensive, and antidiabetic effects [Jolly et al., 2024; Kaur et al., 2024].
Saponins are composed of a sugar moiety (glycone) and an aglycone, also referred to as sapogenin. Their classification is primarily based on the structure of the aglycone – which may have either a triterpenoid or steroidal skeleton – as well as on the number and type of sugar units present in the glycone [Vincken et al., 2007]. The glycone consists of a linear or branched oligosaccharide chain, typically containing hexoses and pentoses such as glucose, galactose, arabinose, rhamnose, or xylose, and may also include uronic acids, most commonly glucuronic acid [Jolly et al., 2024].
The saponin composition and content in plants exhibit substantial variability and depend on multiple factors, including species, plant organ, environmental conditions, and developmental stage. Environmental stressors, such as soil salinity, drought, nutrient deficiencies, and elevated temperatures, can markedly reduce saponin levels [Kaur et al., 2024; Sharma et al., 2022]. In addition, saponin accumulation is strongly species- and growth-stage-dependent: in tea plants, saponin levels increase during early maturation and later stabilize, whereas in yam and Lycium barbarum peak accumulation occurs during tuber development or fruiting stages [Kregiel et al., 2017; Yu & He, 2018]. This highlights the need to consider the biological and environmental context when interpreting saponin variability.
Previous research on B. vulgaris saponins has primarily focused on their composition in mature plants and across different cultivars, whereas information on their variation across different growth stages is currently lacking [Mikołajczyk-Bator et al., 2016a, 2024; Mroczek et al., 2012, 2019, 2021]. The aim of the present study was to investigate organ-, cultivar-, and growth-stage-dependent variation in saponin content in B. vulgaris using liquid chromatography–electrospray ionization-tandem mass spectrometry (LC–ESI-MS/MS), with particular emphasis on seasonal changes in total and individual saponins.
MATERIALS AND METHODS
Solvents and reference compounds
Respective standards from a previous study on B. vulgaris (Red Sphere cultivar) were used for saponin identification [Spórna-Kucab & Wybraniec, 2020], along with extracts of red, yellow, and white B. vulgaris cultivars – Ceryl, Chrobry, Forono, Tytus, and Boldor [Spórna-Kucab et al., 2022]. Oleanolic acid standards were purchased from Sigma-Aldrich (St. Louis, MO, USA). Liquid chromatography–mass spectrometry (LC–MS)-grade acetonitrile and formic acid (purity ≥98%) were obtained from Sigma-Aldrich. Acetone was purchased from Avantor Performance Materials Poland S.A. (Gliwice, Poland). All chemicals and solvents were of analytical grade and used as received. Water utilized throughout the experiments with a resistivity of 18.0 mΩ/cm at 295 K was deionized through a Milli-Q purification system (Merck Millipore, Burlington, MA, USA).
Plant materials
Roots and leaves of Beta vulgaris L. cultivars Round Dark Red, Cylindra, and Snow Ball, as well as leaves of Swiss chard (cv. Rhubarb Chard), were collected from plants grown under standard agronomic conditions in a temperate climate zone on fertile, neutral soil in Zręcin, southeastern Poland, without the application of fertilizers. Seeds of Rhubarb Chard and the white cultivar Snow Ball were purchased from Torseed (Toruń, Poland), while seeds of the red cultivars Cylindra and Round Dark Red were obtained from Legutko (Jutrosin, Poland).
Plants were harvested at two-week intervals from June to September 2024. At each harvest date, plant material was collected from multiple individual plants per cultivar, constituting independent biological replicates. Immediately after harvesting, fresh roots and leaves were washed, weighed, and subjected directly to extraction.
Preparation of plant extracts
The roots and leaves of beets were individually blended in a household blender. A 100-g portion of each sample was extracted by maceration with 400 mL of 50% (v/v) acetone for 30 min at room temperature. The extraction was performed three times using a fresh portion of the solvent for each plant part. After each extraction, the resulting extracts were combined, partially concentrated at 25°C under reduced pressure, and then freeze-dried. Finally, the freeze-dried extracts were weighed and used for further studies on the quantitative and qualitative profile of saponins by the LC–ESI-MS/MS system. All extraction and analytical steps were carried out identically for all biological replicates.
LC–ESI-MS/MS – instrumentation and conditions
Analyses were conducted by means of high-performance liquid chromatography coupled with electrospray ionization tandem mass spectrometry on an LCMS-8030 system (Shimadzu, Kyoto, Japan), equipped with a DGU-20A5R degasser, CBM-20A controller, Nexera LC-20ADXR binary pump, and SIL-20ACXR autosampler, operated via LabSolutions software (version 5.60 SP1, Shimadzu). Chromatographic separation was performed on a Kinetex C18 column (150×4.6 mm, 5.0 μm) with a matching guard column (Phenomenex, Torrance, CA, USA), maintained at 40°C. The flow rate was set to 0.5 mL/min. A binary mobile phase consisting of 2% (v/v) aqueous formic acid (solvent A) and acetonitrile (solvent B) was used. For saponin analysis, the gradient program was as follows: 38–60% B for 15 min, 60–99% B for 1 min, 99% B for 2 min, followed by re-equilibration to 38% B; the total run time was 19 min. For oleanolic acid, the gradient consisted of 20–99% B for 7 min, 99% B for 8 min, and re-equilibration to 20% B, giving a total run time of 19 min.
Tandem mass spectrometry (MS/MS) analysis was performed on a triple-quadrupole mass spectrometer with an electrospray ion source. The following ESI-MS/MS parameters were applied: curved desolvation line (CDL) and heat-block temperatures of 230°C, nebulizing gas flow of 1.5 L/min, electrospray voltage of 4.5 kV, and capillary temperature of 250°C, with nitrogen used as nebulizing and drying gas. Collision-induced dissociation (CID) was carried out with argon as the collision gas, using a collision energy of 35 V. Data were acquired in a negative-ion mode using full-scan (m/z 100–2,000) and selected-ion monitoring (SIM).
Analysis of saponins
Prior to instrumental analysis of saponins, 10 mg of each freeze-dried extract were dissolved in 200 µL of demineralized water and centrifuged at 3,000×g for 5 min using microcentrifuge type 320 (UNIPAN, Warsaw, Poland). Subsequently, 150 µL of the supernatant were collected for further analysis. Aliquots of 15 µL of each sample and 5 µL of the standard solution were injected into the LC–ESI-MS/MS system.
Each extract, originating from an independent biological replicate, was analyzed in three independent technical replicates. Saponin identification was based on characteristic MS/MS fragmentation patterns, accurate mass data, and comparison with data previously reported for B. vulgaris triterpene saponins [Tekieli et al., 2026]. Diagnostic aglycone fragment ions and neutral losses of sugar moieties were used for structural assignment. Individual saponins as well as the total saponin content were quantified using an external calibration curve prepared with oleanolic acid in the concentration range of 1.25–20.0 µg/mL. Oleanolic acid was selected as the calibration standard because it represents the aglycone of the major oleanane-type saponins reported in B. vulgaris, including betavulgarosides, and due to the limited availability of individual saponin reference standards.
The calibration curve, constructed based on MS peak area vs. concentration, exhibited excellent linearity (R2=0.9996). The content of saponins was expressed as mg of saponin per kg of fresh weight of root or leaf material (mg/kg FW). All samples were processed under identical analytical conditions to ensure comparability.
Statistical analysis
All quantitative results are presented as mean and standard deviation (SD) calculated from two independent biological replicates (material collected from different individual plants for each cultivar, organ, and harvest date). Each biological replicate was independently extracted and analyzed in three technical LC–ESI-MS/MS replicates; technical replicates were averaged prior to statistical analysis.
The effects of cultivar, harvest time, and their interaction on the total saponin content and on the contents of individual saponins were evaluated using two-way analysis of variance (ANOVA), performed separately for leaves and roots. When significant main effects or interactions were detected, post hoc multiple comparisons were conducted using Tukey’s honestly significant difference (HSD) test. A significance level of p≤0.05 was applied. Normality of residuals was verified using the Shapiro-Wilk test, and homogeneity of variances was assessed prior to ANOVA.
Principal component analysis (PCA) and hierarchical clustering analysis (HCA) were applied as exploratory multivariate tools to visualize relationships among samples based on the full set of 32 quantified saponins. Statistical analyses were conducted using RStudio (version 2023.13.0+386, Posit Software, PBC, Boston, MA, USA).
RESULTS AND DISCUSSION
Identification of triterpene saponins in B. vulgaris
In the present study, LC–ESI–MS/MS analysis of extracts from four B. vulgaris cultivars (Round Dark Red, Cylindra, Rhubarb Chard, and Snow Ball) revealed a total of 32 triterpene saponins, reflecting substantial diversity in aglycone structures and sugar moieties, including uronic acid (UrA), pentoses (Pen), hexoses (Hex), and substituted acetal (Act) and dioxolane (Diox) residues. The detected saponins comprised oleanane-type, akebonoic acid–based, hederagenin- and gypsogenin-derived aglycones. Because closely related oleanane-type aglycones may generate highly similar MS/MS fragmentation patterns, aglycone assignments based solely on LC–ESI-MS/MS data were discussed at the oleanane-type level unless supported by authentic standards or previously reported nuclear magnetic resonance (NMR) data, particularly in the case of features consistent with GOTCAB-type (glycosides of oleanane-type triterpenoid carboxylic acid bisdesmosides) structures [Arslan, 2017, 2020; Arslan & Cenzano, 2021]. Most compounds were identified using precursor ion masses, MS/MS spectra, and reference standards, while the remaining features were tentatively annotated based on literature fragmentation data. Detailed analytical parameters are summarized in Table 1. Representative chemical structures of the dominant and structurally confirmed saponins identified in the investigated cultivars are shown in Figure 1.
Table 1
Chromatographic and mass-spectrometric data of identified saponins in the Beta vulgaris L. extracts.
[i] Aglycone annotations are based on LC–ESI–MS/MS data. Due to the well-recognized similarity of fragmentation patterns among closely related oleanane-type saponins – including structures consistent with GOTCAB saponins (oleanane-type triterpenoid carboxylic acid 3,28-bisdesmosides) – compounds yielding characteristic oleanane-type fragments are reported at the oleanane-type level unless supported by authentic standards or literature NMR data. For defined betavulgarosides (I–X), names and aglycone identities are discussed in the text based on previously reported NMR evidence. tR, Retention time; m/z, mass-to-charge ratio.
Figure 1
Chemical structures of the dominant saponins in Beta vulgaris L. cultivars: Round Dark Red, Cylindra, Snow Ball, and Rhubarb Chard.
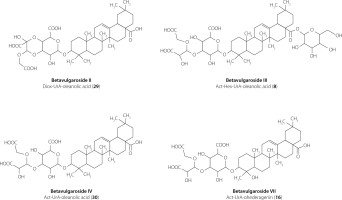
Oleanane-type saponins
Oleanane-type saponins predominated in the saponin profile and represented the most structurally diverse group, comprising 20 detected compounds (Table 1). Oleanane-type cores were detected predominantly in glycosylated form, as indicated by a characteristic neutral loss of 176 Da corresponding to a uronic acid residue [Mroczek et al., 2012, 2019; Spórna-Kucab & Wybraniec, 2020; Wen et al., 2023]. The simplest oleanane-type saponins (31 and 32; m/z 631) showed diagnostic fragments at m/z 455 and a neutral loss of 176 Da, which was consistent with a uronic acid–substituted oleanane core, and were confirmed by comparison with reference compounds previously isolated from B. vulgaris.
The structural diversity of oleanane-type saponins is largely generated by modifications of the core unit composed of an oleanane-type aglycone and uronic acid (m/z 631), which varies through the attachment of additional sugar residues. Accordingly, several structural variants were identified, including mono- and diglycosylated derivatives bearing pentose, hexose, dioxolane, or acetal substituents (saponins 5 and 26–30). Selected structures were confirmed using reference standards (saponins 26–30) or literature data (saponin 5).
Oleanane-type saponins bearing two sugar groups were observed in several configurations, including Hex–Pen (saponins 12 and 19), Diox–Hex (saponin 11), Act–Pen (saponins 21 and 24), and Act–Hex (saponins 8, 17, and 19). Saponins 8, 11, 17, and 19 were confirmed using authentic standards or previously reported NMR data [Yoshikawa et al., 1996], whereas saponins 12 and 19 were confirmed by comparison with reference compounds, and saponins 21 and 24 were assigned based on literature data [Mroczek et al., 2021]. These saponins have been previously reported in B. vulgaris tissues [Mroczek et al., 2012, 2019, 2021; Mikołajczyk-Bator et al., 2016a, 2024].
Triglycosylated oleanane-type saponins were also detected, including Act–Hex–Hex (saponins 3 and 7), Hex–Hex–Hex (saponin 1), and Act–Hex–Pen (saponin 6). An acetal substituent derived from a hexose unit was confirmed for saponins 3, 6, and 7 using reference standards, whereas saponin 1 was tentatively identified based on literature MS data [Mroczek et al., 2012, 2019]. Saponins 3 and 7 (betavulgaroside V) are widely distributed in B. vulgaris cultivars and have also been reported in Achyranthes fauriei [Edelmann et al., 2020a,b; Ida et al., 1995; Mroczek et al., 2012, 2019, 2021; Mikołajczyk-Bator et al., 2016, 2024; Yoshikawa et al., 1998].
Saponins with akebonoic acid aglycone
In addition to oleanane-type saponins, B. vulgaris contained triterpene saponins with akebonoic acid as the aglycone, identified by a diagnostic fragment ion at m/z 439 under negative ESI conditions (Table 1). The simplest derivative, saponin 25 (m/z 615), was assigned as an akebonoic acid–uronic acid conjugate based on a characteristic neutral loss of 176 Da from the C-3 position of the aglycone and represents a compound not previously reported in B. vulgaris.
Single-glycosylated akebonoic acid derivatives were detected as saponin 20 (m/z 747; pentose substituent), tentatively assigned based on its MS/MS fragmentation pattern, and saponin 22 (m/z 777; acetal substituent), which was unambiguously identified by comparison with reference standards. More complex derivatives included saponin 14 (m/z 909; betavulgaroside X), identified based on sequential sugar losses and previously reported NMR data [Yoshikawa et al., 1998], as well as saponins 4 and 13 (m/z 939; betavulgaroside VIII), both confirmed using reference standards and previously reported in B. vulgaris and related beet cultivars [Edelmann et al., 2020a,b; Mikołajczyk-Bator et al., 2016; Mroczek et al., 2021; Yoshikawa et al., 1998].
Hederagenin and gypsogenin aglycones of saponins
B. vulgaris contained a minor group of triterpene saponins with hederagenin and gypsogenin aglycones (Table 1), detected at much lower levels than oleanane- and akebonoic acid–based derivatives and characterized by diagnostic ions at m/z 471 and 469, respectively. Saponin 18 (m/z 647) was identified as a hederagenin monoglycoside bearing a uronic acid residue based on a neutral loss of 176 Da and has not been previously reported in B. vulgaris. Compounds 10, 15, and 16 (m/z 779 and 809) were identified as hederagenin-based saponins containing a uronic acid residue and additional hexose and/or pentose units or an acetal substituent, as confirmed by a diagnostic fragment at m/z 471. Saponin 16 (betavulgaroside VII) is the most thoroughly characterized saponin in this group, with its structure elucidated by NMR [Yoshikawa et al., 1998] and reported across numerous B. vulgaris cultivars [Mikołajczyk-Bator et al., 2016a,b, 2024]. Saponins 15 and 16 were confirmed using reference standards, whereas saponin 10 was assigned based on literature data [Mroczek et al., 2012; Mikołajczyk-Bator et al., 2016a,b, 2024].
Saponin 2 (m/z 969) was identified as a hederagenin-based saponin bearing a uronic acid, a hexose, and a dioxolane substituent and has previously been reported only in sugar beet cultivars [Mikołajczyk-Bator et al., 2016b]. Saponin 23 (m/z 807) was the sole gypsogenin-based saponin detected, as confirmed by a diagnostic fragment at m/z 469, and was identified by comparison with reference standards; this compound has been previously reported in sugar beet and Swiss chard cultivars [Mikołajczyk-Bator et al., 2016b; Mroczek et al., 2021].
Analytical precision and variability assessment
Instrumental precision and extraction repeatability were evaluated using three independent samples prepared from the same homogeneous plant material and subjected to the complete extraction procedure. Each resulting extract was analyzed three times by LC–ESI-MS/MS to assess instrumental variability.
The relative standard deviation (RSD) obtained from repeated LC–ESI-MS/MS analyses of the same extract (n=3) ranged from 2.7% to 4.3%, indicating satisfactory instrumental precision. Extraction repeatability was assessed by comparing the results of three independent extractions performed on identical plant material. The RSD values calculated between these extractions ranged from 2.3% to 9%, which was considered acceptable given the complexity of plant matrices. Biological variability was assessed based on the two independent biological replicates collected for each cultivar, organ, and harvest date and expressed as biological RSD (%). The biological RSD values exhibited a relatively broad range. For individual saponins, biological RSD values generally ranged from approximately 5% to 16%, with the majority of compounds showing variability between 7% and 14%, depending on cultivar, organ, and harvest time.
The relative standard deviation (RSD) values for total saponins (calculated as the sum of individual saponins for each biological replicate) most commonly fell within the range of approximately 8–11%, although slightly higher values were occasionally recorded at specific harvest dates or for particular cultivars (Tables S1–S8 in Supplementary Materials).
The reduced variability of the total saponin content reflects the cumulative nature of this parameter, whereby variability in individual saponins partially compensates when summed at the level of biological replicates. In cases where a given saponin was not detected in either biological replicate (mean value equal to zero), calculation of RSD was not applicable and was therefore omitted.
Saponin quantitative analysis
The total saponin content in leaves and roots of B. vulgaris cultivars harvested at different times is shown in Figure 2, and the relative proportions of the 32 identified compounds in Figure 3. Additionally, the contents of individual saponins in the fresh weight of the plant materials are presented in Tables S1–S8. Quantitative LC–ESI-MS/MS analysis demonstrated that saponin accumulation in Beta vulgaris L. was influenced by plant organ, harvest date and cultivar. Two-way ANOVA showed highly significant effects of cultivar, harvest time, and their interaction on total saponin content in leaves (f=137.73 for cultivar, f=68.26 for time, f=25.50 for cultivar × time; all p<0.0001) and in roots (f=77.45, 69.36, 31.56, respectively; all p<0.0001). Similarly strong effects were observed for most individual saponins (typically p<0.0001). Detailed two-way ANOVA outputs for individual saponins and total saponins in leaves and roots are provided in Table S9 in Supplementary Materials.
Figure 2
Total saponin content in leaves (A) and roots (B) of four Beta vulgaris L. cultivars across seven harvest dates. Data are expressed as mean and standard deviation. Different lowercase letters indicate significant differences among cultivars within each harvest date (Tukey’s HSD test, p≤0.05). FW, fresh weight.
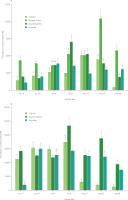
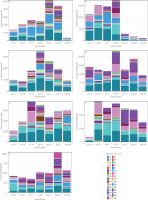
Figure 3
Stacked bar plots showing the total content and relative proportions of 32 saponins in leaves and roots of four Beta. vulgaris L. cultivars collected across seven harvest dates. Panels: (A) Cylindra leaves, (B) Cylindra roots, (C) Round Dark Red leaves, (D) Round Dark Red roots, (E) Snow Ball leaves, (F) Snow Ball roots, and (G) Rhubarb Chard leaves. Bar height represents total saponin content, and colors indicate individual saponins S1–S32, which are listed in Table 1. FW, fresh weight.
Organ-dependent quantitative trends in total saponin content
Across cultivars and harvest dates, roots accumulated higher total saponin levels than leaves, with a much wider range of values (Figure 2 and Figure 3). Total saponin content ranged from 386 to 10,414 mg/kg FW in leaves and from 1,170 to 23,298 mg/kg FW in roots. These values are consistent with those reported previously [Spórna-Kucab et al., 2022], where up to 20,810 mg/kg FW were found in beetroot peels of various cultivars. Although Mroczek et al. [2019] reported higher total saponin levels in leaves for some cultivars, their study also demonstrated that, depending on genotype, roots may accumulate equal or even higher concentrations. Beyond the overall differences between organs, distinct temporal patterns were also evident. Seasonal dynamics were organ-specific: total saponin content in leaves peaked in mid-to-late season (depending on cultivar), whereas roots exhibited a pronounced mid-season maximum followed by a decline (Figure 2 and Figure 3). This pattern likely reflects developmental regulation of secondary metabolism and tissue-specific biosynthesis, with preferential accumulation in protective outer tissues. Indeed, Spórna-Kucab et al. [2022] demonstrated significantly higher saponin concentrations in beet peel compared to the flesh, supporting their enhanced deposition in tissues directly exposed to environmental stressors.
Cultivar-specific patterns of total saponin accumulation
In leaves, genotype-related differences were pronounced and strongly dependent on harvest date. The cultivar Rhubarb Chard showed the highest total saponin content, reaching 10,414 mg/kg FW on 25 August. The highest value for the cultivar Round Dark Red was recorded on 28 July (7,010 mg/kg FW), for the cultivar Cylindra on 11 August (5,060 mg/kg FW), and for the cultivar Snow Ball on 14 July (3,861 mg/kg FW) (Figure 2 and Figure 3). These results are consistent with Mroczek et al. [2019], who demonstrated significant variability in saponin content among B. vulgaris genotypes.
Root tissues also displayed clear genotype-dependent accumulation patterns. The greatest accumulation capacity was observed for the cultivar Round Dark Red, which reached 23,290 mg/kg FW on 28 July. The cultivar Cylindra exhibited a pronounced mid-season maximum (17,330 mg/kg FW on 28 July), followed by a sharp decline to 1,170 mg/kg FW on 8 September. The cultivar Snow Ball showed elevated early-to-midseason total saponin content (14,910 mg/kg FW on 30 June), gradually decreasing to 7,310 mg/kg FW by 8 September (Figure 2 and Figure 3). These trends are consistent with previous reports; Spórna-Kucab et al. [2022] documented approximately 20,810 mg/kg in the peel of yellow beet cultivar Boldor, compared with only 500 mg/kg in the flesh of white Snow Ball, indicating strong tissue-specific distribution. Likewise, Mikołajczyk-Bator et al. [2024] reported significantly higher saponin content in root skins than in the flesh. Such findings support the mid-season maxima observed here and likely reflect preferential accumulation in outer protective tissues, followed by dilution or metabolic redistribution at later developmental stages.
Quantitative variability of individual saponins
Individual saponins showed pronounced heterogeneity in abundance as well as in temporal and cultivar-dependent patterns (Figure 3, Tables S1–S8). Many low-abundance compounds were detected only intermittently, whereas a limited number of high-abundance saponins dominated the overall profile, including in particular, betavulgarosides II (29), III (8), IV (30), VII (16) and Act-UrA-akebonoic acid saponin (22). These major compounds exhibited coordinated seasonal variation in both temporal occurrence and abundance, including cultivar-dependent maxima. Two-way ANOVA showed highly significant effects of cultivar, harvest date, and their interaction on these and most other saponins (p<0.0001), supporting the idea of a differentiated biosynthetic network (Table S9). This aligns with Mroczek et al. [2019], who described various accumulation rates of specific saponins and spatially differentiated biosynthesis across organs.
Principal component and hierarchical clustering analyses
PCA performed on the dataset comprising 32 saponins clearly separated the samples according to plant organ (Figure 4). The first principal component (PC1), explaining 24% of the total variance, distinctly discriminated root samples (positive PC1 values) from leaf samples (negative PC1 values), while the second principal component (PC2), accounting for 17% of the variance, further differentiated samples according to harvest date.
Figure 4
Principal component analysis (PCA) score plot of saponin profiles in leaves (circles) and roots (squares) of four Beta vulgaris L. cultivars collected across seven harvest dates. PC1 explains 24% and PC2 explains 17% of the total variance. Colors indicate cultivars.
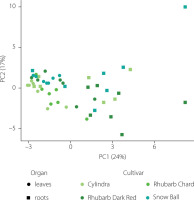
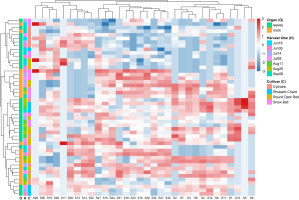
The separation along PC1 and PC2 was primarily driven by four high-loading saponins, corresponding to betavulgaroside III (8), betavulgaroside VII (16), Act–UrA–akebonoic acid saponin (22), and betavulgarosides II (29) and IV (30), previously described in beet [Mroczek et al., 2019]. Their consistent contribution across samples indicates that these compounds constitute the principal sources of variability within the dataset. Hierarchical clustering analysis (heatmap visualization) fully supported the PCA results. Samples were grouped primarily according to plant organ (root vs. leaf), followed by harvest date and cultivar, revealing a clear underlying structure in saponin distribution patterns (Figure 5).
Figure 5
Hierarchical clustering and heatmap of 32 saponins detected in leaves and roots of different Beta vulgaris L. cultivars across seven harvest dates. The heatmap presents normalized relative liquid chromatography–mass spectrometry signal intensities (z-scores) of individual saponins across all samples (red = higher relative abundance, blue = lower relative abundance). Rows correspond to samples and columns to individual saponins. Colored side bars indicate harvest date (time), plant organ (leaves or roots), and cultivar. Dendrograms represent hierarchical clustering of samples (left) and saponins (top), illustrating similarities in saponin profiles and co-variation patterns among compounds.
Biological significance of triterpene saponins identified in B. vulgaris
The triterpene saponins identified in B. vulgaris constitute a structurally diverse and biologically relevant group of secondary metabolites with well-established functional and pharmacological importance. The predominance of oleanane-type saponins observed in this study suggests that this compound class may play a central role in determining the biological and functional properties of beet-derived tissues and products. In particular, the presence of NMR-confirmed betavulgarosides [Yoshikawa et al., 1996, 1998], previously associated with anti-inflammatory, hepatoprotective, antimicrobial, antioxidant, and anticancer activities, underscores their potential contribution to the health-promoting effects of B. vulgaris [Castellano et al., 2022; Günther & Bednarczyk-Cwynar, 2025; Gupta, 2022; Lisiak et al., 2021; Liu et al., 2019; Sen, 2020; Yoon & Choi, 2010].
The biological activity of oleanolic acid–based saponins is strongly influenced by their glycosylation patterns. The presence of uronic acid residues together with additional hexose or pentose units modulates key physicochemical properties, including solubility, membrane interactions, and bioavailability [Francis et al., 2002]. The predominance of glycosylated oleanane-type cores detected in the present study is consistent with earlier reports indicating that biologically-active forms of oleanolic acid occur predominantly as saponins rather than as free aglycones in plant tissues [De Tommasi et al., 1991; Kinjo et al., 1999].
Beyond oleanane-type compounds, the identification of akebonoic acid–based saponins further expands the biological relevance of the saponin profile of B. vulgaris. Akebonoic acid, a 30-noroleanane triterpenoid, has been reported to exhibit pronounced anticancer, antibacterial, and antidiabetic activities [Dirir et al., 2021; Wang et al., 2014]. Although the biological functions of its glycosylated derivatives remain insufficiently characterized, their detection in beet tissues suggests that B. vulgaris may represent a valuable source of structurally-unique saponins with largely unexplored bioactivity. The occurrence of simple monoglycosidic derivatives may additionally reflect early biosynthetic intermediates or compounds with enhanced membrane-disrupting potential [Böttger & Melzig, 2013].
Minor saponins containing hederagenin and gypsogenin aglycones further contribute to the functional complexity of the saponin mixture in B. vulgaris. Both aglycones are known for their cytotoxic, antimicrobial, antifungal, and antiviral properties [Cheng et al., 2018; Ciftci et al., 2018; Engholm et al., 2017; Favel et al., 1994; Krasteva et al., 2014; Su et al., 2024; Sun et al., 2023; Xie et al., 2023; Zeng et al., 2018]. Despite their lower abundance compared to oleanane- and akebonoic acid–based saponins, these compounds may still be biologically-relevant due to potential additive or synergistic interactions within complex saponin assemblies.
Taken together, the diversity of aglycone scaffolds and glycosylation patterns observed in B. vulgaris highlights this species as a rich source of triterpene saponins with multifaceted biological potential. The tissue-specific and season-dependent variability in saponin composition demonstrated in this study may have important implications for the nutritional value, functional characteristics, and potential health benefits of beet-derived products. Nevertheless, further investigations employing purified compounds and targeted bioassays are required to establish direct links between saponin composition, concentration, and biological activity.
CONCLUSIONS
This study demonstrates that saponin accumulation in Beta vulgaris L. is strongly dependent on plant organ, cultivar, and harvest time, with significant interaction effects between these factors. Roots accumulated higher total saponin levels than leaves and exhibited a pronounced mid-season maximum followed by a decline, whereas leaf saponin contents generally peaked in mid-to-late season depending on genotype.
Quantitative variability was largely driven by a limited group of dominant saponins, which showed coordinated seasonal and cultivar-dependent dynamics. Multivariate analyses confirmed that plant organ represents the primary source of variance in the dataset, with additional structuring according to harvest date and genotype.
Overall, the results indicate that saponin biosynthesis in B. vulgaris is developmentally-regulated and spatially-differentiated between organs. From a practical perspective, both cultivar selection and optimized harvest timing are critical parameters for maximizing total saponin yield and shaping the qualitative composition of beet-derived plant material.

