INTRODUCTION
Nigella (Nigella sativa L.) is an annual plant native to regions including Western Asia, the Middle East, Central Europe, and North Africa. The plant produces aromatic seeds of an intense black color, commonly known as black cumin. Since antiquity, N. sativa seeds have gained centuries-old recognition as a high-quality spice for their distinct aroma and have also been used for therapeutic purposes in several countries around the world [Ahmad et al., 2021]. Lipids and essential oils derived from these seeds have long been valued as natural remedies for treating a wide range of illnesses, including asthma, hypertension, diabetes, and influenza [Majeed et al., 2021]. N. sativa seeds contain various natural compounds, including lipids, polysaccharides, proteins, flavonoids, alkaloids, and saponins [Ahmad et al., 2021]. The lipid fraction represents the major group among these organic constituents, accounting for 30–40% of the total seed weight [Ahmad et al., 2021]. This designation qualifies N. sativa seeds as an excellent oilseed when compared to other vegetable matrices such as olive, soybean, and sunflower. Vegetable matrices, especially oleaginous fruit and seeds, contain a complex mixture of lipids consisting of glycerides (approximately 98%) and unsaponifiable matter (approximately 2%) [Sakouhi et al., 2011]. The glyceridic fraction of vegetable oils comprises mainly triacylglycerols esterified with saturated, monounsaturated, and polyunsaturated fatty acids [Dubois et al., 2007]. The lipid fraction of vegetable matrices equally contains numerous unsaponifiable compounds, such as sterols, triterpenic alcohols, vitamins, and policosanols, all of which exhibit notable biological activities. Sterols and triterpenic alcohols constitute the major portion of the unsaponifiable fraction in vegetable oils [Goriainov et al., 2021]. The term policosanols refers to a mixture of long-chain (C20–C36) aliphatic primary alcohols. Weerawatanakorn et al. [2019] reported that tetracosanol (C24), hexacosanol (C26), octacosanol (C28), and triacontanol (C30) were the main policosanols in oleaginous matrices. Policosanols have been shown to be associated with numerous physiological benefits, such as platelet aggregation inhibition, endothelial damage mitigation, and a cholesterol-lowering effect [Weerawatanakorn et al., 2019]. The unsaponifiable fraction also contains tocopherols, which are well known for their effective antioxidant properties [Patterson, 1981]. These natural compounds protect lipids from peroxidation by neutralizing lipid peroxyl radicals or interacting with singlet oxygen and other reactive oxygen species. They have a crucial role in preventing lipid oxidation, a major form of degradation that can occur during food processing, distribution, storage, and preparation.
In many food industries, protection against spoilage and rancidity is commonly achieved using synthetic antioxidants, including ascorbyl palmitate, butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), and propyl gallate (PG). Nonetheless, concerns have increasingly emerged regarding the potential toxic and carcinogenic effects associated with these synthetic additives [Wang et al., 2021]. As a result, there is growing demand within these industries for natural antioxidants (tocopherols and phenolic compounds) extracted from vegetable matrices. Such additives can be incorporated directly into food products to preserve them against alteration and rancidity. Beyond their antioxidant activity, many plant extracts also contain antimicrobial active compounds that could enhance the storage stability of food products [Karnwal & Malik, 2024]. As far as microbial damage is concerned, most food industries typically rely on synthetic antimicrobial agents, such as benzoates, sorbates, propionates, and nitriles for food preservation from microbial damage. However, several studies have highlighted that these chemical preservatives may exhibit potential toxic, mutagenic, or genotoxic effects, raising growing safety concerns [Piper & Piper, 2017]. Recent reviews have emphasized the need to replace such synthetic compounds with safer, naturally occurring antimicrobial alternatives [Karnwal & Malik, 2024]. These natural additives do not only have the advantage of preserving food products against alterations, rancidity, and microbial infections but also offering nutritional benefits to consumers.
Recently, food industry has started exploring new oil plant matrices with both nutritional and preservative potential. Most of these bioactive compounds are primarily extracted from plant sources, such as chia, flax, pumpkin, sunflower, and sesame seeds [Alasalvar et al., 2021; Sumara et al., 2023]. However, research on the bioactive lipid compounds of N. sativa seeds, particularly the unsaponifiable fraction, remains scarce [Albakry et al., 2022]. To the best of our knowledge, this is the first study to provide a comprehensive profile of both unsaponifiable matter and phenolic compounds from N. sativa seeds and to discuss their potential applications in nutrition and food preservation. The objective of this research is twofold: first, to identify and characterize the bioactive lipid compounds in N. sativa seeds, such as unsaturated fatty acids, phytosterols, policosanol, and tocopherols; and second, to assess their antioxidant and antimicrobial activities of their seed extracts, thereby supporting the potential future applications of N. sativa seeds.
MATERIALS AND METHODS
Reagents and standards
Chloroform, diethyl ether, ethanol, methanol, n-hexane, and isopropyl alcohol (high-performance liquid chromatography, HPLC, grade) were obtained from Panreac Applichem ITW Reagents (Darmstadt, Germany). Anhydrous sodium carbonate (Na2CO3), Folin–Ciocalteu (FC) reagent, aluminum chloride (AlCl3), sodium nitrite (NaNO2), anhydrous sodium sulfate (Na2SO4), and potassium hydroxide (KOH) were purchased from VWR (Leuven, Belgium). A standard mixture of fatty acid methyl esters, N,O-bis(trimethylsilyl)trifluoroacetamide (BSTFA), 5α-cholestanol and 1-eicosanol standards, α-, β-, γ-, and δ-tocopherol isoform standards, gallic acid (GA), quercetin, hydrochloric acid, (+)-catechin standard, 1,1-diphenyl-2-picrylhydrazyl (DPPH) radicals, and butylated hydroxytoluene (BHT) standard were supplied by Sigma-Aldrich (Madrid, Spain). 2,4,6-Tris(2-pyridyl)-s-triazine (TPTZ) was purchased from Fluka (Buchs, Switzerland). Mueller–Hinton agar was obtained from BIOKAR Diagnostics (Allonne, France).
Material
Samples of Nigella sativa L. seeds were collected in June 2023 from four distinct geographic regions of Tunisia: Nabeul, Kairouan, Sfax, and Gabès. The cultivation of N. sativa in Tunisia was managed and supervised by the Ministry of Agriculture. These regions display a variety of climatic conditions and geographic coordinates: (1) Nabeul: longitude: 36°26’N; latitude: 10°43’E; altitude: 14 m; annual rainfall: 404 mm/year; (2) Kairouan: longitude: 10°05’N; latitude: 10°05’E; altitude: 68 m; annual rainfall: 300 mm/year; (3) Sfax: longitude: 34°45’N; latitude: 10°25’E; altitude: 13 m; annual rainfall: 233.1 mm/year; and (4) Gabès: longitude: 33°49’ N; latitude: 9°45’ E; altitude: 1 m; annual rainfall: 152 mm/year.
The N. sativa seeds were hand-harvested randomly, and 2 kg of seeds approximately were collected from each region. The seeds collected from each site were ground separately into a fine powder and preserved at 4°C for further analysis.
Extraction of fixed oil from N. sativa seeds
Oil extraction from N. sativa seeds was performed using the Folch method, following the procedure described in our previous publication [Sakouhi et al., 2023]. The extraction of oils was performed using a chloroform/methanol mixture (1:1, v/v), with 5 mL per 1 g of ground seeds. After extraction, the solvent was removed under reduced pressure using a rotary evaporation. The obtained N. sativa seed oil was weighed and stored in dark bottles at 4°C for analysis of fatty acids, phytosterols, policosanol, and tocopherols.
Determination of fatty acid composition of Nigella sativa L. seed oil
The fatty acid profile of N. sativa seed oil was determined using the method described by Sakouhi et al. [2023]. The fatty acid methyl esters from each sample were extracted by vigorously shaking a mixture of N. sativa oil in n-hexane (0.2 g in 3 mL) with 0.4 mL of 2 M methanolic solution of potassium hydroxide. A 1 μL aliquot of the solution was then injected into a gas chromatograph (GC) equipped with a flame ionization detector (FID) (Hewlett-Packard, Avondale, PA, USA). The analysis was achieved using a fused silica column (50 m length × 0.25 mm i.d.) coated with SGL-1000 phase (0.25 μm thickness; Sugerlabor, Madrid, Spain). Helium served as the carrier gas at a constant flow rate of 1 mL/min. The injector and detector temperatures were set at 250°C, while the oven temperature was maintained at 210°C. Fatty acids were identified by comparing their retention times to those of a standard fatty acid methyl ester mixture. The fatty acid (FA) relative content (expressed as % of total fatty acids) was calculated using Equation (1):
where: PAi is FA’s peak area and TPA is total FA peak area. The mean value of three injections was reported.
Saponification of oil and thin layer chromatography separation of phytosterols and policosanols
The unsaponifiable fraction was obtained by saponifying 5 g of N. sativa seed oil from each location using 50 mL of a 12% (w/v) ethanolic KOH solution following the procedure we used previously [Sakouhi et al., 2023]. The mixture was heated at 60°C for 1.5 h, and the unsaponifiable compounds were then extracted four times with 50 mL of petroleum ether. After extraction, the obtained unsaponifiable matter was separated into subfractions using preparative thin-layer chromatography (TLC) on silica gel 60 G F254 plates (Merck, Darmstadt, Germany) with hexane and diethyl ether mixture (6:4, v/v) as a developing solvent [Sakouhi et al., 2023]. To accurately identify the sterol and policosanol bands among the other unsaponifiable subfractions, reference solutions of 5α-cholestanol and 1-eicosanol (external standards) were applied to the TLC plates. After chromatographic development, the plate was sprayed with 2,7-dichlorofluorescein and examined under UV light. Subsequently, the phytosterol and policosanol bands were scraped off, extracted three times with a chloroform and diethyl ether mixture (1:1, v/v), filtered to eliminate residual silica, and finally dried with a rotary evaporator. The final extracts were derivatized separately with BSTFA and then stored at −4°C until further analysis by gas chromatography–mass spectrometry (GC-MS).
Gas chromatography-mass spectrometry analysis of phytosterols and policosanols
The analysis of phytosterols and policosanols of N. sativa seed oil was conducted according to the GC-MS method [Sakouhi et al., 2023], on a Varian SAR 3400CX gas chromatograph connected directly to a Varian SATURN mass detector (Varian, Palo Alto, CA, USA). A DB-5MS fused silica capillary column (30 m × 0.25 mm i.d., 0.25 µm film thickness; J&W Scientific, Folsom, CA, USA) was used for the separation. Helium was used as the carrier gas at a constant flow rate of 1 mL/min. The injector and detector temperatures were maintained at 250°C. The oven temperature was programmed to increase from 150°C to 300°C at a rate of 4 °C/min, with the final temperature held for 10 min. The transfer line temperature was also set at 250°C. Mass spectra were recorded using electron impact ionization at 70 eV. A 1 μL aliquot of each sample was manually injected in the split mode with a split ratio of 60:1. Phytosterols and policosanols were identified by comparing their retention times and mass spectra with those of authentic standards, and further confirmed using the NIST/EPA/NIH Mass Spectral Library, NIST 2020 version (accessed November 2024). The quantification of phytosterols and policosanols was performed through internal standard methods using 5α-cholestanol and 1-heneicosanol, respectively, and the results for individual phytosterols were expressed in mg/100 g of oil and those for policosanols in mg/kg of oil. The analysis was performed in triplicate.
High-performance liquid chromatography analysis of tocopherols from N. sativa seed oil
Tocopherol analysis of N. sativa seed oil was conducted following the American Oil Chemists’ Society (AOCS) method Ce-8-89 [AOCS, 1989]. In brief, 0.5 g of oil was dissolved in n-hexane in a 5-mL volumetric flask. The resulting solution was then filtered through a 0.45-μm polytetrafluoroethylene membrane filter, and 20 μL of the filtrate was injected into an Agilent 1200 Series HPLC system (Agilent Corp., Santa Clara, CA, USA), equipped with a Phenomenex Luna Sil column (250 × 4.6 mm i.d., 5 μm particle size; Phenomenex, Inc., Torrance, CA, USA). The column was maintained at 40°C throughout the analysis. The mobile phase consisted of a mixture of n-hexane and isopropyl alcohol (99:1, v/v). It was used at a flow rate of 1.0 mL/min. Prior to use, the mobile phase was degassed by sonication for 10 min. Fluorescence detection was performed at an excitation wavelength of 290 nm and at an emission wavelength of 330 nm. Tocopherol peaks were identified by comparing their retention times to those of external standards of pure α-, β-, γ-, and δ-tocopherol isoforms. The quantification of tocopherols in N. sativa oil samples was carried out using the external standard calibration method. Standard solutions of each tocopherol isomer (0.1 mg/mL) were prepared in n-hexane. Calibration curves were obtained by plotting the peak area against the corresponding standard concentrations at four calibration levels, and quantification was based on the resulting linear regression equations. The analysis was performed in triplicate, and results were expressed as mg/100 g oil.
Preparation of different solvent extracts of N. sativa seeds
N. sativa seeds were extracted using three different solvents: ethanol, a water and ethanol mixture (20/80, v/v), and water, using the maceration method. For each solvent, 5 g of seed powder from each location was macerated in 50 mL of the solvent for 24 h at room temperature, with continuous agitation at 150 rpm. The resulting filtrates were dried and then stored at 4°C until further analysis of their antioxidant and antibacterial activities.
Determination of total phenolic content of the extracts
The total phenolic (TP) content of the different N. sativa seed extracts (aqueous, hydroethanolic, and ethanolic) was determined using the method of Singleton & Rossi [1965]. Briefly, 63 µL of FC reagent were added to 63 µL of each extract at a concentration of 0.01 g/mL. After a 5-min incubation period, 625 µL of a 7% (w/v) Na2CO3 solution were added, and the mixture was incubated in the dark for 90 min. After incubation, 200 µL of each mixture were transferred to a microplate and analyzed with a Synergy HTX MultiMode Microplate Reader (Biotek Instruments, Winooski, VT, USA) at a wavelength of 765 nm. The total phenolic content was calculated using a standard calibration curve for gallic acid with a concentration range of 10–100 mg/mL. The linear regression equation obtained was: y = 8.1722x + 0.0308, with an R2 of 0.9974. The TP content was expressed as mg of gallic acid equivalents (GAE) per g of dry extract. Measurements for each extract were performed in triplicate.
Determination of total flavonoid content of the extracts
The total flavonoid (TF) content of the N. sativa seed extracts (aqueous, hydroethanolic, and ethanolic) was determined using the method with AlCl3, as defined by Zhishen et al. [1999]. In short, 38 µL of 5% (w/v) NaNO2 were added to 125 µL of each extract (0.01 g/mL). The mixture was allowed to stand for 6 min, after which 75 µL of freshly prepared 10% (w/v) AlCl3 solution were added. After incubation, 200 µL of each mixture were transferred to a microplate and analyzed with a Synergy HTX MultiMode Microplate Reader (Biotek Instruments) at a wavelength of 510 nm. TF content was calculated relying on a standard calibration curve for quercetin with concentrations of 10–75 mg/mL. The linear regression equation was: y = 0.0103x + 0.0943, with an R2 of 0.9996. The TF content was expressed as mg of quercetin equivalents (QE) per 100 g of dry extract. Measurements were carried out in three experimental replicates for each extract.
Determination of total tannin content of the extracts
The total tannin (TT) content of the N. sativa seed extracts (aqueous, hydroethanolic, and ethanolic) was determined using the method described by Julkunen-Tiitto [1985]. Briefly, 50 µL of each extract (0.01 g/mL) were mixed with 1.5 mL of 4% (v/v) vanillin in methanol and 750 µL of a 12 M hydrochloric acid solution. The mixture was thoroughly blended and incubated in the dark at room temperature for 20 min. After incubation, 200 µL of each mixture was transferred to a microplate and analyzed with a Synergy HTX MultiMode Microplate Reader (BioTek Instruments) at a wavelength of 500 nm. The TT content was calculated using a (+)-catechin standard calibration curve with concentrations of 0.10–1 mg/mL. The linear regression equation was: y = 0.8962x + 0.0014, with an R2 of 0.9995. The TT content was expressed as mg of (+)-catechin equivalents (CE) per g of dry extract. Measurements were taken in triplicate for each extract.
Evaluation of the antioxidant activity of N. sativa seed extracts
DPPH assay
The antioxidant activity of each N. sativa seed extract was evaluated using the DPPH assay by Brand-Williams et al. [1995]. A DPPH radical methanol solution (0.35 g/L), previously adjusted to an absorbance of 0.95 at 515 nm with methanol, was placed into the wells of a microplate at a volume of 190 µL. Stock solutions (0.1 mg/mL) of the extracts were prepared in their respective extraction solvents (ethanol, water, and ethanol-water (20:80, v/v)), while the BHT stock solution (0.1 mg/mL) was prepared in methanol. These solutions were then diluted to obtain five different concentrations in a range of 10–100 µg/mL for extracts and 5–25 µg/mL for BHT. Subsequently, 10 µL of each diluted extract and BHT solution or each solvent alone was added to the wells with DPPH radical solution, and the mixtures were incubated in the dark for 50 min at room temperature. After incubation, the absorbance was read. The inhibition percentage (I%) was calculated using Equation (2):
where: Acontrol is the absorbance of the DPPH radical solution without the antioxidant and Asample is the absorbance of the DPPH radical solution with the extract or BHT.
The antioxidant activity was quantified by the IC50 value (expressed as mg/mL), which represents the concentration of an extract or BHT required to reduce 50% of the DPPH radicals. Measurements were performed in triplicate for each sample.
Ferric-reducing antioxidant power assay
The ferric-reducing antioxidant power (FRAP) of the extracts was determined according to the method of Benzie & Strain [1996], with slight modifications. The FRAP reagent was freshly prepared before each analysis by mixing 10 mL of acetate buffer (300 mM, pH 3.6), 1 mL of a TPTZ solution (10 mM in 40 mM HCl), and 1 mL of an FeCl3×6H2O solution (20 mM). The mixture was maintained at 37°C until use. In a 96-well microplate, 25 µL of extract solutions (1 mg/mL) or Trolox standard solutions (20–200 mg/L) were mixed with 175 µL of the pre-warmed FRAP reagent. The absorbance was recorded at 593 nm and the antioxidant power was expressed as Trolox equivalents per g of dry extract weight (mg TE/g dry extract) based on the linear standard curve with R2=0.9976. Measurements were performed in triplicate for each sample.
Determination of the antibacterial activity of N. sativa seed extracts
In this part of the study, only the ethanolic extracts of N. sativa seeds from various locations were evaluated for antibacterial activity, as the aqueous and hydroethanolic extracts showed no significant activity against the tested bacterial strains. The ethanolic extracts were tested against four bacterial strains commonly found in food products: three Gram-negative bacteria (Escherichia coli ATCC 8739, Pseudomonas aeruginosa ATCC 27853, and Salmonella enteritidis ATCC 13076) and one Gram-positive bacterium (Staphylococcus aureus ATCC 6538). These bacterial strains were sourced from the American Type Culture Collection (ATCC, Manassas, VA, USA), and the analyses were conducted at the Laboratory for Microbiological Analysis of Food Products, part of the National Laboratory for Analysis and Testing in Tunis, Tunisia. Antibacterial activity was assessed using the agar well diffusion method with sterile Mueller–Hinton agar, as described by Weerakkody et al. [2010]. Specifically, a 100 µL aliquot of a freshly prepared bacterial suspension (adjusted to 107 CFU/mL) was inoculated onto the surface of the agar plates. Sterile filter paper discs (6 mm in diameter) were impregnated with 20 µL of N. sativa seed extract (10 mg/mL) and 20 µL of gentamycin (10 mg/mL), a synthetic antibiotic used as a positive control, and placed on the agar plates. The plates were incubated at 37°C for 24 h. Subsequently, the microbial inhibition was assessed by measuring the diameter (mm) of the inhibition zone (DIZ, clear zone around each disc). Measurements were carried out in triplicate.
Statistical analysis
All data were analyzed using the XLSTAT software package (Addinsoft, NY, USA) for Microsoft Excel. Significant differences in the parameters analyzed among the samples of N. sativa seeds from four different Tunisian regions were evaluated using one-way analysis of variance (ANOVA) with a post-hoc Tukey test, with a significance level set at p<0.05.
RESULTS AND DISCUSSION
Fatty acid composition of N. sativa seed oil
The lipid content of the studied N. sativa seeds was 23.41, 25.32, 27.15, and 28.64 g/100 g, respectively, for seeds collected from the Nabeul, Kairouan, Sfax, and Gabès regions. Fatty acids are the main constituents of the lipid fraction and contribute specific physicochemical characteristics to fats and oils. The fatty acid profile of each N. sativa seed oil was determined using GC-FID, with the results summarized in Table 1. Nine compounds were identified. Among these, linoleic acid (C18:2) was the most abundant, accounting for 49.66% to 58.47% of the total fatty acids in all samples. The high linoleic acid content, an essential ω-6 fatty acid, suggests that N. sativa oil could serve as a valuable source of this vital fatty acid, which plays an important role in regulating cholesterol levels [Azemi et al., 2023]. Additionally, oleic acid (C18:1), the second most prevalent fatty acid, accounted for 19.63% to 29.25% of the total fatty acids in the samples. Thus, it contributed a substantial proportion of monounsaturated fatty acids in N. sativa seed oil. This acid is well known for its cardioprotective benefits and positive impact on lipid metabolism [Petersen et al., 2024]. Nevertheless, palmitic (C16:0), stearic (C18:0), eicosadienoic (C20:2), linolenic (C18:3), eicosanoic (C20:0), and eicosenoic (C20:1) acids were found in lower contents across all samples. While these fatty acids are present in smaller amounts, they play an essential role in maintaining the structural integrity of cell membranes [Pashkovskaya et al., 2018]. The results also revealed significant differences (p<0.05) in the contents of the major fatty acids across the four regions, with linoleic acid being the highest in Gabès, followed by oleic acid in Nabeul and palmitic acid in Gabès. These variations are likely to be due to the distinct agro-climatic conditions in the studied regions, as they differ considerably in terms of annual rainfall, which ranges from 404 mm in Nabeul to 152 mm in Gabès. Similar results regarding the fatty acid composition of N. sativa seed oil have been reported by Kiani et al. [2020], who identified the same fatty acids, albeit with slight differences in their relative percentages.
Table 1
Fatty acid composition (expressed as % of total fatty acids) in Nigella sativa L. seed oils from four Tunisian regions (Nabeul, Kairouan, Sfax, and Gabès).
The results of the present study demonstrate that N. sativa seeds share a fatty acid composition comparable to that of chia, flax, pumpkin, sesame, and sunflower seeds, which are widely marketed for their health benefits [Ahmed et al., 2026; Sumara et al., 2023]. Our findings highlight N. sativa seeds as an equally valuable source of bioactive fatty acids, with linoleic and oleic acids as the major components.
Phytosterol composition of N. sativa seed oil
Phytosterols represent the primary constituents of the unsaponifiable fraction found in vegetable oils and are recognized for their nutritional significance in the human diet. In this study, the sterol fraction of N. sativa oil was separated from the total unsaponifiable matter using TLC (Figure 1) and subsequently characterized through GC-MS. The identified phytosterols and their respective contents are summarized in Table 2. A total of eight sterol compounds were detected in N. sativa seed oils, which were classified into two structural categories. The first category, Δ5-sterols, includes cholesterol, campesterol, stigmasterol, β-sitosterol, Δ5-avenasterol, and Δ5,24-stigmastadienol. The second category comprises Δ7-sterols, specifically Δ7-stigmastenol and Δ7-avenasterol. Quantitative assessment showed that β-sitosterol was the most abundant phytosterol across all oil samples. The highest content of β-sitosterol was found in the seeds collected from Nabeul in northern Tunisia (100.46 mg/100 g oil), while the lowest was observed in the samples from Gabès in the southern region (88.19 mg/100 g oil). Additionally, other sterols such as stigmasterol (ranging from 40.23 to 54.21 mg/100 g oil), campesterol (27.52 to 33.76 mg/100 g oil), and Δ5-avenasterol (16.60 to 22.42 mg/100 g oil), were consistently present at relatively high levels in seed oils across all regions. The content of the main phytosterols in seeds from different regions exhibited significant variation. This difference can primarily be attributed to agro-climatic factors, as the four studied regions (Nabeul, Kairouan, Sfax, and Gabès) exhibit significantly different climatic conditions. Comparable profiles of phytosterols in N. sativa oil have been documented by Albakry et al. [2022], although slight variations in their contents were noted.
Table 2
Phytosterol composition (expressed in mg/100 g of oil) in Nigella sativa L. seed oils from four Tunisian regions (Nabeul, Kairouan, Sfax, and Gabès).
Figure 1
Thin-layer chromatography (TLC) separation of phytosterols and policosanols of the unsaponifiable fraction of Nigella sativa L. seed oil.
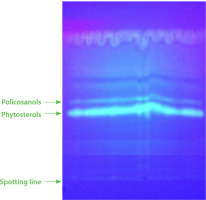
Phytosterols have attracted growing scientific attention due to their beneficial effects on human health. Evidence from clinical research highlights their potential in reducing cholesterol levels, as well as their anti-inflammatory and anticancer properties [Jiménez et al., 2024]. For example, a study by Trautwein et al. [2018] demonstrated that phytosterol intake led to triglyceride reductions ranging from 0.8% to 18%, with the greatest reductions observed in individuals who had elevated baseline triglyceride levels. Given their cholesterol-lowering properties in mammals, soluble phytosterols have been incorporated into various food products. Current dietary recommendations suggest an average phytosterol intake of approximately 250 mg/day for healthy nutrition [Nattagh-Eshtivani et al., 2022]. Comparing the phytosterol profiles of seeds like chia, flax, pumpkin, and sesame [Alasalvar et al., 2021], N. sativa seeds exhibit a similar composition, with β-sitosterol, stigmasterol, campesterol, and ∆5-avenasterol as the primary compounds. Thus, N. sativa seeds represent a valuable plant-based source of phytosterols, offering nutritional and health benefits, and can be classified as functional food seeds.
Policosanol composition of N. sativa seed oil
The analysis of the policosanol composition in N. sativa seed oils revealed the presence of eleven distinct compounds including eicosanol (C20), docosanol (C22), tetracosanol (C24), pentacosanol (C25), hexacosanol (C26), heptacosanol (C27), octacosanol (C28), nonacosanol (C29), triacontanol (C30), hentricontanol (C31), and dotriacontanol (C32) (Table 3). Among these, hexacosanol, octacosanol, triacontanol, and dotriacontanol were the major policosanols of N. sativa seed oil, accounting for more than 65% of the total policosanol content. Triacontanol was the most abundant, with contents of 30.48, 27.74, 25.36 and 24.13 mg/kg oil of seeds from Nabeul, Kairoun, Sfax, and Gabès, respectively. In contrast, policosanols with an odd number of carbon atoms (pentacosanol, heptacosanol, and hentricontanol) were detected in lower amounts.
Table 3
Policosanol composition (expressed in mg/kg of oil) in Nigella sativa L. seed oils from four Tunisian regions (Nabeul, Kairouan, Sfax, and Gabès).
The total policosanol content of N. sativa seed oil (average 90 mg/kg) placed it among other natural, rich sources of these bioactive compounds; when compared with previously reported values, this content was substantially higher than that found in brown beeswax (5.2 mg/kg) and yellow beeswax (12 mg/kg) [Jackson et al., 2006], and it also exceeded the 17.4 mg/kg reported for whole sugar cane. Although sugar cane leaves and peels remain richer sources (181 and 270 mg/kg, respectively) [de Lucas et al., 2007], the values obtained for N. sativa seeds oil indicate that these seeds are also a valuable and underutilized plant material in this respect.
Tocopherol composition of N. sativa seed oil
Tocopherols are exclusively synthesized by photosynthetic organisms and primarily act as antioxidants. They play a critical role in improving the oxidative stability of vegetable oils and enhancing the nutritional value of crop plants for human consumption. The tocopherol isomers identified in N. sativa seed oils (α, β, γ, and δ) and their respective contents in the samples from the four regions are presented in Table 4. The data indicate that α-tocopherol was the most abundant isomer, with relatively high contents ranging from 10.38 to 11.51 mg/100 g oil across all seed samples. γ-Tocopherol was also found in significant amounts, ranging from 7.38 to 8.45 mg/100 g oil. Conversely, β-tocopherol and δ-tocopherol were detected in smaller amounts, ranging between 1 and 2 mg/100 g oil, across the seeds from all regions. These results are consistent with those reported by Albakry et al. [2022] for N. sativa seeds obtained from an Iranian supermarket, which showed a comparable distribution of tocopherol isomers.
Table 4
Tocopherol isomer composition (expressed mg/100 g of oil) in Nigella sativa L. seed oils from four Tunisian regions (Nabeul, Kairouan, Sfax, and Gabès).
Tocopherols act as antioxidants because they can neutralize lipid-free radicals by donating a hydrogen atom from their phenolic group. In fact, it was reported that the hydrogen-donating capacity of tocopherol isomers follows the order: α>β>γ>δ [Kamal-Eldin & Appelqvist, 1996]. Additionally, Patterson [1981] demonstrated that one molecule of tocopherol can protect the oxidation of 103 to 106 molecules of unsaturated fatty acids. Natural α-tocopherol has been shown to effectively inhibit lipid oxidation, thereby improving the oxidative stability of lipid-rich food products during storage, often performing comparably or even better than certain synthetic antioxidants, such as tertiary butylhydroquinone (TBHQ) [Alizadeh et al., 2019]. This underscores the further use of N. sativa seed oil, a valuable source of these lipophilic tocopherols, as a food additive.
Total phenolic, total flavonoid, and total tannin contents of N. sativa seed extracts
The TP content of the different extracts (aqueous, ethanolic, and hydroethanolic) of N. sativa seeds ranged from 60.24 to 74.35 mg GAE/g dry extract across the four studied localities (Table 5). The highest amount was recorded in the hydroethanolic extract, ranging from 72.5 mg GAE/g (seeds from the Nabeul region) to 74.35 mg GAE/g (seeds from Gabès). These findings highlight the crucial role of solvent type and polarity in the extraction efficiency of total phenolic content. In fact, the water-ethanol mixture proved most effective for extracting phenolics due to the complementary polarities of the solvents, whereas distilled water and pure ethanol were less efficient, extracting only highly polar or moderately polar compounds, respectively. The results obtained using maceration were higher than those reported by Mariod et al. [2009], who found that the TP content of N. sativa seed extracts from Malaysia was 27.8 mg GAE/g of dry extract when methanol was used as the sonication solvent. However, using a Soxhlet apparatus, Zwolan et al. [2020] reported TP contents of 24.89 and 35.72 mg GAE/g of dry extract when water and ethanol were used as extraction solvents, respectively.
Table 5
Total phenolic (TP), total flavonoid (TF), and total tannin (TT) contents of extracts of Nigella sativa L. seeds from four locations obtained using various solvents – ethanol, 80% (v/v) ethanol, and water.
[i] Values are mean ± standard deviation, n=3. Values for each parameter (TP, TF and TT) followed by the different lowercase letter (a–c) in the same row (origin comparison) or uppercase letters (A–C) in the same column (solvent comparison) are significantly different (p<0.05). GAE, gallic acid equivalents; QE, quercetin equivalents; CE, (+)-catechin equivalents.
The total flavonoid content of N. sativa seed extracts ranged from 17.19 to 29.47 mg QE/100 g, depending on the extraction solvent used and the sampling region (Table 5). Similar to determining TF content, the ethanol-water mixture proved to be the most effective solvent for flavonoid extraction (yielding the highest amount 29.47 mg QE/100 g in dry extract of seeds from the Gabès region), followed by ethanol, and finally distilled water. However, the difference in TF content between the hydroethanolic and ethanolic extracts was less obvious in this case compared to TP content. Ravi et al. [2024] recorded a TF content of 7.31–8.15 mg QE/100 g for Indian cumin seeds. Ouattar et al. [2022] found that the methanol-water extract of Moroccan N. sativa seeds contained 25.8 mg QE/100 g of total flavonoids when maceration was used as the extraction method. These variations in total flavonoid content among N. sativa seeds can be attributed to several factors, including genotypic and environmental differences within species, the sampling period, and the analytical methods used for quantification.
The total tannin content of the different extracts (aqueous, ethanolic, and hydroethanolic) of N. sativa seeds ranged from 4.13 to 10.19 mg CE/g dry extract across the four studied localities (Table 5). As with total phenolics and flavonoids, the hydroethanolic solvent exhibited the highest extraction efficiency for total tannins. This can be attributed to the strong solubility of tannins, both hydrolyzable and condensed, in polar solvents, particularly water-alcohol mixtures. Similar to total phenolic (TP) and total flavonoid (TF) contents, N. sativa seeds from the Gabès region exhibit the highest total tannin content, regardless of the extraction solvent used. This is because N. sativa produces more secondary defense metabolites, including flavonoids, tannins, alkaloids, and thymoquinone, when exposed to a hot climate, enhancing its resilience and adaptability to these challenging conditions [Ibrahim et al., 2023].
Antioxidant activity of N. sativa seed extracts
In the present study, the antioxidant potential of N. sativa seed extracts was evaluated using two complementary analytical assays: DPPH radical scavenging activity and ferric-reducing antioxidant power. The antioxidant activity of the seed extracts was expressed as IC50 and mg TE/g dry extract for the DPPH and FRAP assays, respectively (Table 6). The results indicated that the hydroethanolic extracts exhibited significantly (p<0.05) higher antioxidant activity compared to the ethanolic and aqueous extracts, regardless of the assay employed. The DPPH IC50 values for the hydroethanolic extracts of N. sativa seeds collected from the Nabeul, Kairouan, Sfax, and Gabès regions were 3.25, 3.10, 2.93, and 2.82 mg/mL, respectively. Previous study by Gueffai et al. [2022] showed that the DPPH IC50 value of the hydroethanolic (5:5, v/v) extract of N. sativa seeds was 1.96 mg/mL when obtained by maceration, whereas it decreased to 1.14 mg/mL under optimized ultrasound-assisted extraction conditions. The FRAP of N. sativa seed extracts ranged from 187 to 819 mg TE/g across the different sampling locations (Table 6). The lowest values were determined for the aqueous extracts (187–316 mg TE/g), whereas the intermediate (588–724 mg TE/g) and highest (646–819 mg TE/g) values were obtained when ethanol and aqueous ethanol were used as solvents, respectively. The highest FRAP (819 mg ET/g) was recorded in the hydroethanolic extract of N. sativa seeds from the Gabès region. Kadam & Lele [2017] reported that delipidated N. sativa seeds from India exhibited a FRAP of 1.85 mM TE/g (equivalent to 463 mg TE/g) when extracted with an ethanol-water (60:40, v/v). Overall, the FRAP obtained in the present study using ethanol-water (80:20, v/v) was higher than this reported by Kadam & Lele [2017]. This difference may be explained by the fact that during lipid extraction process, some secondary metabolites such as, phenolics may migrate into the oil fraction, thereby reducing the antioxidant potential measured in the remaining sample.
Table 6
Antioxidant activity determined in DPPH assay (expressed as IC50, mg/mL) and ferric-reducing antioxidant power (FRAP) (expressed as mg Trolox equivalent, TE/g dry extract) of various solvent extracts of Nigella sativa L. seeds from four Tunisian regions (Nabeul, Kairouan, Sfax and Gabès).
[i] Values are mean ± standard deviation, n=3. Values followed by the different lowercase letter (a–c) in the same row or uppercase letters (A–C) in the same column (separately for each assay) are significantly different (p<0.05). IC50, the concentration required to inhibit 50% of the DPPH radicals. The butylated hydroxytoluene (BHT) was used in DPPH assay as reference antioxidant, with an IC50 value of 0.46 mg/mL.
According to the experimental results, the hydroethanolic solvent has proven to be more effective at extracting maximum antioxidant compounds from N. sativa seeds, including phenolics, which are secondary metabolites known for their antioxidant properties. The ethanol effectively extracts also thymoquinone, a prominent biomolecule characteristic of N. sativa seeds [Ahmad et al., 2021]. This bioactive compound has been shown to exhibit a wide range of therapeutic properties, including antioxidant, anti-inflammatory, antihistamine, antibacterial, anticancer, and encephalomyelitis effects in various in vitro and certain in vivo studies [Akter et al., 2021; Alberts et al., 2024; Kazemi et al., 2024].
Antimicrobial activity of N. sativa seed ethanolic extract
Table 7 shows the inhibition zone diameters for the ethanolic extracts of N. sativa seeds from different locations, alongside those for gentamicin, a reference antibiotic, tested against four foodborne pathogens. The results indicate that all the examined bacterial strains exhibited sensitivity to the ethanolic extracts of N. sativa seeds. The diameter of the inhibition zone varied from 11.25 to 18.43 mm, depending on the bacterial strain and the region where the N. sativa seeds were harvested. Notably, the S. aureus strain demonstrated the highest sensitivity to the ethanolic extract, with inhibition zones ranging from 16.74 to 18.43 mm, compared to gentamicin which had an inhibition zone of 22.52 mm. This notable sensitivity can be attributed to S. aureus being a Gram-positive bacterium, which lacks an outer membrane, allowing the extract’s compounds to penetrate more easily and inhibit its growth. In contrast, the strains S. enteritidis (14.52 to 16.44 mm), E. coli (13.84 to 15.07 mm), and P. aeruginosa (11.25 to 12.32 mm) exhibited varying levels of sensitivity to the ethanolic extract of N. sativa seeds. The obtained results also indicate that, for the same bacterial strain, the extract from N. sativa seeds harvested in the Gabès region demonstrated the most significant antibacterial effect, with inhibition diameters of 18.43 mm for S. aureus, 16.44 mm for S. enteritidis, 15.07 mm for E. coli, and 12.32 mm for P. aeruginosa. This result suggests that climatic conditions also influence the antibacterial potency, given the significant climatic differences among the regions studied. The accumulation of secondary metabolites is likely to explain the pronounced antibacterial activity of N. sativa seeds from the Gabès region compared to those from the remaining areas. In the literature, few studies address the antimicrobial activities of N. sativa seeds. Consistent with Bourgou et al. [2012], who demonstrated antibacterial activity of methanolic seed extracts against S. aureus and E. coli, our results also show notable inhibition of these strains by ethanolic extracts. Similarly, the antifungal properties reported by Nadaf et al. [2015] and the broad-spectrum antibacterial and antifungal activity of N. sativa oils documented by Tiji et al. [2021] corroborate the view that this seed is a rich source of bioactive compounds with antimicrobial potential. Importantly, our study demonstrates that the antibacterial potency of N. sativa seeds varies with geographic origin, with seeds from the Gabès region showing the strongest inhibitory effects. This suggests that environmental stressors such as high temperature, aridity, and solar radiation in Saharan regions enhance the accumulation of secondary defense metabolites, thereby boosting antimicrobial efficacy. These findings do not only highlight their consistency with earlier studies, but also the fact that agro-climatic conditions shape the bioactivity of N. sativa, reinforcing its potential as a natural antimicrobial source for food preservation and safety.
Table 7
Antimicrobial activity measured by the diameter of the inhibition zone (mm) of ethanolic extracts (10 mg/mL) of Nigella sativa L. seeds from four Tunisian regions (Nabeul, Kairouan, Sfax, and Gabès).
| Region | Escherichia coli | Salmonella enteritidis | Pseudomonas aeruginosa | Staphylococcus aureus |
|---|---|---|---|---|
| Nabeul | 13.84±0.03d | 14.52±0.04d | 11.25±0.02d | 16.74±0.04d |
| Kairouan | 14.24±0.02c | 15.62±0.01c | 11.55±0.04c | 17.63±0.03c |
| Sfax | 14.61±0.04b | 15.81±0.06b | 11.94±0.02b | 17.84±0.02b |
| Gabès | 15.07±0.03a | 16.44±0.05a | 12.32±0.04a | 18.43±0.02a |
| Gentamycin* | 18.30±0.13 | 20.23±0.02 | 16.43±0.02 | 22.52±0.02 |
CONCLUSIONS
The study findings indicated that the N. sativa seeds are rich in bioactive lipids, including fatty acids, phytosterols, policosanols, and tocopherols, confirming their value as a functional food resource. The seed extracts showed notable antioxidant activity while ethanolic extracts particularly exhibited significant antibacterial activity against key foodborne pathogens. Extracts of seeds from the Gabès region displayed the strongest activity, a finding that underscores the influence of harsh Saharan climatic conditions on the accumulation of secondary defense metabolites. Overall, these findings highlight that N. sativa seeds hold great potential for future applications, both in food preservation, as a natural alternative to synthetic additives, and as a valuable source of health-promoting compounds in the human diet. Future research should focus on isolating and characterizing the specific bioactive molecules responsible for these effects, clarifying their mechanisms of action, and testing their applications in food formulations and clinical settings. Such studies will further consolidate the position of N. sativa seeds as a valuable natural resource for nutrition, health, and food safety.

